COVID 19 Vaccine – WHO Action and Inaction
April 3, 2022
COVID 19 Vaccine
–
WHO Action and Inaction
Padmini Arhant
The latest action by the Geneva based World Health Organization (WHO) against Indian manufactured COVAX – the corona virus vaccine by India’s Bharat Biotech suspending exports of Indian vaccine abroad citing inadequate compliance on good manufacturing practice in facility may be one aspect of WHO guidelines and criteria.
“Good manufacturing practices are the practices required in order to conform to the guidelines recommended by agencies that control the authorization and licensing of the manufacture and sale of food and beverages, cosmetics, pharmaceutical products, dietary supplements and medical devices.”
However, WHO as the international body maintaining similar policy for all across geographic locations beginning with inspection of Wuhan lab, Hubei Province, China conducting life endangering biohazard research could have averted catastrophic global pandemic.
WHO and other international committee monitoring and inspection of the Wuhan bio lab and others elsewhere are imperative for science and mankind which evidently was not done at least prior to the onset of the deadly pathogen.
WHO discretionary exercise with Wuhan Virology Institute, China could have saved the world and humanity at large from monumental tragedy and health disaster.
In the same context, United States and western partners funded biochemical labs in non-western parts of the world such as Ukraine, China and other regions allowing independent open investigation to rule out bio endangerment to mankind is an urgent requirement. The world deserve to be free from any and all kinds of life consuming experiments and endeavors in the aftermath of the COVID 19 unleashed pandemic.
As for WHO, the international health organization implementing the same gold standard on good manufacturing practice for manufacturers and suppliers in Europe and the United States where serious ethical and scientific protocol negligence have occurred with no immediate response or action from the domestic or international regulatory body would clarify WHO autonomy and uniformity.
The case in point is the European (Germany) big Pharma Pfizer’s mRNA vaccine clinical trial outsourced to Ventavia Research Group in 2021.
The revelation on Ventavia Research Group as Pfizer’s outsourced research company verifiably engaged in crucial data integrity issues besides ignoring scientific protocol and good practice in the critical Pfizer’s mRNA vaccine clinical trial beckon authorities interest and necessary intervention to protect lives now and in the future.
Despite alarming details from the whistle blower as former employee of Ventavia Research Group followed up British Medical Journal and New England Journal of Medicine articles in this regard,
The vaccine availability for children in specified age group, pregnant women, booster shot etc. proceeded without the matter resolved by WHO, United States FDA, CDC and other agencies responsible for public health and safety.
There was another incident with Johnson & Johnson’s corona virus vaccine notably with sub-contracted production line in Baltimore, Maryland, United States in March 2021.
Courtesy: The New York Times.March 31, 2021.
Factory Mix-Up Ruins Up to 15 Million Vaccine Doses From Johnson & Johnson
“Workers at a plant in Baltimore manufacturing two coronavirus vaccines accidentally conflated the ingredients several weeks ago, contaminating up to 15 million doses of Johnson & Johnson’s vaccine and forcing regulators to delay authorization of the plant’s production lines.”
In light of these flagrant violations in different settings, WHO role as an international regulatory authority applying the rule on fair and equal compliance in the pharmaceutical industry starting with medical research facilities as well as manufacturing and production centers worldwide without exception would be a refreshing change in the otherwise selective interference.
The international and domestic regulations on pharma products and services especially their outsourced agents and sub-contractors handling production units of vaccine and other pharmaceutical products subject to stringent conformity leaving no room for lapses with uninformed inspections would provide element of trust among disillusioned public and concerned whistle blowers risking their career and life in health care industry.
Any update on preventive measures and action taken on 2021 Pfizer vaccine clinical data breach brought to relevant authorities notice such as FDA in the United States, Pfizer and Ventavia Research Group actually involved in the matter would address legitimate grievances expressed by whistle blowers and health professionals not barring general public expectation on transparency and accountability.
Padmini Arhant
Author & Presenter
PadminiArhant.com
Prakrithi.PadminiArhant.com
Pfizer Documents
Comments
Got something to say?
You must be logged in to post a comment.
-
Recent Posts
- US Navy Blockade of Strait of Hormuz
- US – Iran Peace Talks
- US – Iran Ceasefire
- United States – Senate Vote on Federalizing Election Process
- Indian Panorama
- United States’ Objectives on Iran War
- God’s Chosen People – What does it mean?
- Israel and United States War Against Iran
- srael and United States War Conclusion against Iran
- Nation is Indispensable not the transition power
The Wonder Nature - Not created by human. Wise not to destroy it with human invented bombs and nuclear might.
OM Nama Sivaya – Glory to Almighty God!
My Life – Trilogy V1 Author Padmini Achintya Arhant
GOSPEL TRUTH
GOSPEL TRUTH
“You will have enemies and their recruits – the wannabes and opportunists in collusion with a clique.
Here’s why that’s a good thing.
Their obsession with you is proof you are already above them.”
Absolute Truth.
Padmini Arhant
——————————————————————————————
“https://youtu.be/lauiDVF_u04”
“People who hate you usually do it for three reasons.
1. They see you as a threat.
2. They hate themselves.
3. They want to be you.
So don’t take their hate personally.
Hmmm Just keep being yourself.
And Keep moving forward.”
Thank you.
Well said. Couldn’t agree more.
Padmini Arhant
Graceful Exit Exemplify Dignity
Graceful Exit Exemplify Dignity
Padmini Arhant
When your time and term is long over in politics, film industry and media…exit gracefully rather than insist, persist and impose yourself on those against their will.
Anything pushed and exerted is desperation demanding unworthy attention further adding repulsion and rejection.
Above all, spare politics from celluloid screen fiction and fakery eliminating the farcical film industry, Indian in particular, predominantly used for propaganda.
Adolf Hitler, would be turning in his grave for being surpassed in this trade as the trend setter. The result is an established fact.
Politics is real deal involving people’s lives, public money and their destiny – all of which gambled and squandered for power quest leaving the world in shambles as nobody’s business.
Stop using phony media and anchors with AI image.
That goes for deploying endless proxies, puppets and pawns. The strategy is nothing more than platitude that never had any meaningful substance nor real value.
These preliminary measures would sanitize politics from severe contamination.
Your dignity is in your hands. How you treat yourself and others in life.
BTW – where were you all especially those relentlessly vying and competing in holier than thou contest?
Haven’t you all been around over decades even as farther back 1970, 80, 90 and 2000 onward?
Did it not occur to any of you about your speciality as Messiah and Diva – that you are flaunting now in adherence to your recruiters’ monetized script?
Were there no epiphany and revelation about you?
The difference between fraud and factual is crystal clear like that of darkness and light.
Sun and moon never advertise their presence unlike celebrity syndrome famished for eternal free publicity.
Padmini Arhant
Nigeria / India
Fact Check – Controversial ICE Created in 2012 by Democrat Presidency Barack Obama
Fact Check – Controversial ICE Created in 2012 by Democrat Presidency Barack Obama
ICE was introduced and implemented in 2012 by democrat Presidency Barack Obama.
The democrat Presidency then deployed ICE and deported over 2 million Hispanics including documented workers having conned and scammed the Hispanic vote block with citizenship promise that was never delivered.
Instead Latinos were deported in sheer betrayal of voter TRUST after securing their votes in the hard to win 2012 Presidential re-election.
In fact, with African American electorate disillusionment in the first term of the Black Presidency, it was the Hispanic vote in 2012 that facilitated to the finish line on ballots count.
Yet, soon after the election, the re-elected democrat Barack Obama Presidency pulled the rug under the feet of the same Latinos in a typical Barack Obama – use and abuse exploitative strategy.
Furthermore, the 2012 was pre-arranged to cover all bases in mutual pact with then Republican contender Mitt Romney in name only to cede election drowning the Republican votes in that election.
Barack Obama Presidency proudly launched the most undemocratic rule National Defense Authorization (NDA) in 2010 – 2014. NDA was even worse than predecessor Bush – Cheney’s Patriot Act.
Under Barack Obama executed NDA in 2009 – 2016 term, it politically targeted American citizens on American soil for trials in military tribunal and not civilian courts, the norm by far in direct violation of Constitutional rights.
The same Constitution derided by the democrat Presidency Barack Obama as nothing more than a “Piece of Parchment.”
The source of ICE, NDA etc., are now on the other side of the fence with criticism having initiated and institutionalized the controversial agencies abusing power and authority.
Not all Presidential crimes could be wiped out with RACE card used as political impunity with no end in sight on criminality long after expiry of term in office in 2016.
Padmini Arhant
Israel’s Mummification of (late) PM Benjamin Netanyahu
Israel’s Mummification of (late) PM Benjamin Netanyahu
Has Israel and India’s Narendra Modi made similar arrangement for other Israeli top brass from Mossad’s Chief to Cabinet Minister, IDF head, Benjamin Netanyahu’s brother, besides late Benjamin Netanyahu wiped out in Iranian missiles in early March 2026?
Or,
The AI optics and Narendra Modi’s 👍🏾 reserved only for deceased PM Netanyahu?
Alternatively, did Iran’s missile only struck all of the above mentioned at the meeting seated next to each other under the same roof sparing Benjamin Netanyahu’s life as unique anointed by the likes of Narendra Modi and league?
The concerted effort to keep the lie alive by Indian PM Narendra Modi and alike, is the same old leaving the foot in the mouth with Indian PM Narendra Modi ever starved for publicity reminiscence of making Chai (tea) from the sewage gas navigating towards political status quo.
Food for thought – Perhaps Indian PM Narendra Modi’s nuska (tip) cooking from the sewage gas could come in handy with India currently facing gas and fuel shortage to cater for 1.5 billion population.
When thousand lies to cover up irrefutable reality on life and death and anything is the norm for such politics,
There is no doubt speaking the Truth is not Narendra Modi’s cup of tea.
Padmini Arhant
Israel’s Conundrum
Israel precision elimination of top brass leaders, officials in Iran, Hamas and Hezbollah pose a major credibility issue.
Where was Israel prior to and on October 7, 2023 missing in action to avert Hamas terror attack?
The event subsequently used as the premise to wage war against Gaza in October 2023 and now in Iran 2025 – 2026.WHY IS ISRAEL NOT PRESENTING THE DECEASED PM BENJAMIN NETANYAHU UNTIL NOW BARRING AI OPTICS?
Israel’s latest serial killing of Iran’s security chief Ali Larijani is to avenge revenge of all top leaders in Israel beginning with the killing of PM Benjamin Netanyahu, Mossad Chief, Israeli cabinet minister and IDF head…instant deaths in Iran’s missile strike few days ago.
Israel could track foreign leaders movement anyhow and somehow leading to assassinations, however remained clueless on October 7, 2023 Hamas, always on Israel’s radar sudden disappearance leading to October 7, 2023 in Israel is a conundrum for Israeli intelligence and political authority.
Padmini Arhant
Berserk Mind Expedite Self-Termination – G7 has Kenya in Megalomania
Berserk Mind Expedite Self-Termination – G7 has Kenya in Megalomania
G-7 excludes South Africa and replaced by East African Kenya – Barry Soetoro aka Barack Obama Native land.
Kenya induction is megalomania. Similar to street and public square in Northern and Southern California named after Barack Obama – having done nothing except exploit, extract and extort Californians using democrat State agencies to bully, harass and unlawfully collect money in penalty and interests on over taxed payments paid long before due date.
The devil gone berserk with the brain affected since age 8 years from hard substance revealed in all of these bizarre developments.
The devil is no longer in disguise unravelled in these dramatic sequences.
When the END is upon the devil wreaking havoc, the berserk mind expedite Self-Termination.
Padmini Arhant
தமிழ் நாட்டின் பொது தல பிரச்சாரம் என்ற விபச்சாரம்
தமிழ் நாட்டின் பொது தல பிரச்சாரம் என்ற விபச்சாரம்
முதலில் தொப்பியை கழட்டு. பிறகு திராணி தைரியம் இருந்தால் “ஜீ’ பட்டம் போடாமல், மற்றும் கறுப்பு, சிவப்பு। என்ற நிற முகமுடியை கிழித்து – அயோக்கியர்கள், அராஜகம் அட்டூழியம் அவர்களின் பிறவி குணம் மற்றும் செயல்களாயின், அதன்படி விமர்சனத்தை வை.
ஏனென்றால், நிறமல்ல அறம் மற்றுமே முக்கியம். அதே போல் இனமல்ல மனம் குணம் தான் மனிதனின் தன்மைபும், செயல்பாட்டையும் உன்னதப் படுத்துகிறது.
கறுப்பு, சிகபபு என்பதை பார்க்காமல் – அவன் நடத்தை, செயல்களை கவனி. போலி, கல்லம், கபடம், கல்மிஷம், நயவஞ்சகம்…அது தான் ஒருவரின் அடையாளம்.
அதை விட்டுட்டு, காசை வீசினால் நாலு வேசியாக இந்த பிரச்சார தொழில் – ஸோஷியல் மீடியாவை விபச்சாரமாக மாற்றி அநீதியை பராமரிக்கும் நரி கும்பலாக இந்த காட்டு மிராண்டிகளுக்கு அவர்கள் போடும் பிச்சைக்கு நன்றி கடனாக உண்மையை மறைப்பதும், பொய்யை மெய்யாக்கும் கேவலமான தொழில் தனக்கும், யார் சார்பில் இவ்வாறு நடப்பதோ, ஒட்டு மொத்த அனைவருக்கும் அழிவு மற்றும் இழிவாகும்.
தன் வினை தன்னைச் சுடும், உரட்டியப்பன் வீட்டைச் சுடும்.
அதற்க்கும் மேல் –
அரசன் அன்று கேட்டால், பாவக் கடலில் மூழ்கி தினறும் இந்த கலியுகத்தில், தெய்வம் இன்றே கொல்லும்.
பத்மினி அர்ஹந்த்
Name with the vowel ending in ‘i’ and phonetic ‘y’
Name with the vowel ending in ‘i’and phonetic ‘y’
In the contemporary topsy turvy world due to twisted politics, the obsession with the name ending in vowel ‘i’ or the phonetic ‘y’ is targeted by nomadic minds that regards the names with ‘i’ and ‘y’, their adversary and arch nemesis.
Not to mention, the names starting with ‘P’ served dime a dozen by desperadoes in the fake and make-believe charade.
So what’s up with that bizarre addiction to defeat and / or eliminate those with that name?
Regardless, desperadoes propose and God disposes – in the resolute will with original remaining immutable.
In 2012, it was then Republic contender Mitt Rom(ney), incentivized to cede election before hand in favor of the democrat incumbent Barack Obama confronted with abysmal prospects in 2012 re-election following shellacking in mid-term congressional election in 2010.
Israel’s fixation translated in pursuits with names ending in ‘i’ and / or phonetic ‘y’.
Israel’s assassinated members in Iran.
IRGC Commander and Military Officer – Qasem Solei(mani) in 2020.
Iran’s Supreme leader – Ayatollah Ali Khame(nei) and family on February 28, 2026.
IRGC Senior military officer and Commander, Basij Forces – Gholamreza Solei(mani).
Iran’s top security official – Ali Larija(ni).
Gaza war – 2023 – 2026.
Israel assassinated Hamas leaders.
Among key Hamas officials assassinated by Israel,
Hamas chief Ismail Ha(ni)yeh in 2024.
Lebanon.
Israel assassinated Hezbollah members.
The exception in this instance yet based on commonality with Iran – Palestine Independence.
Hassan Nasrallah, the secretary-general of Hezbollah, was assassinated in an Israeli airstrike in Beirut in September 2024.
From Barack Obama to Israel – the obsession targeting names with (ni), (mi) and phonetic ‘y’ is compulsive disorder and fanaticism driving the obsessed to imminent self-destruction experienced by them and witnessed by the world.
Accordingly, Iran selecting leaderships with names not ending in vowel (i) in (ni), (mi) and phonetic ‘y’ would perhaps deter the fad among assassins turning them into rabid morbid killers in the predator instinct.
Padmini Arhant
Fuel Crisis and Alternatives to the World at Large
Fuel Crisis and Alternatives to the World at Large
The fuel crises confronting world population due to Israel and United States war against Iran leaving the Middle East in turmoil is effectively war against humanity.
Israel and United States war tied entirely to gain full control over oil and energy resources besides strategic dominance is no different from the COVID pandemic unleashed for selective few western profitability risking human lives and health with irreversible loss in the aftermath of millions of deaths worldwide in the pandemic.
The nature endowments are thankfully not limited to natural oil & gas in the world. The other power generators through myriad sources such as Solar, wind, hydro (water) and nuclear power projects facilitate electricity / power production.
Utilizing these natural elements to the optimum would significantly reduce oil & gas dependency, the crux of war mongering consuming innocent lives and civilians in every warfare.
As a matter of fact, alternative energy and renewable energy maximum utilization not just during the war fueled crisis, ordinarily customizing the practice world over would have direct impact containing resource related and dominance warfare.
World population usage of electric and hybrid power, solar power etc., would mitigate shortage of gas and oil supply.
In daily household requirements, electric cooking units from Microwave to Conventional wall oven, electric cooking pot, rice cooker, coffee makers, baking units are available online and in retail stores in most parts of the world.
Similarly, electric and hybrid vehicles for limited radius miles and kilometers would provide energy reserves for major vehicle operation like trucks and airplanes as well as industrial consumption.
Those who have these mod-cons using them to maximum at present and other times would spare the gas and oil for households and even businesses dependent on these fuels to operate without having to shut down or even fall victim to price gouging ( exponential hike in prices of essential goods) taking advantage of the ongoing crisis from warfare to exert hegemony against Iran and throughout Middle East.
Energy Saving Options: Car pool beginning with family and commuters to work – using one vehicle for allowed capacity rather than one per vehicle would save enormous fuel.
Similarly using regular public transport besides electric trains within city and areas of operation also aid in fuel conservation. Youth riding their bikes using bike lanes that is necessary in thoroughfare and busy traffic is overall good for health and natural environment.
Intelligence without “common sense” is nonsense.
Padmini Arhant
இநதியா என்ன கெஸ்,எரி பொருள் கட்டுப்பாடா? கவலை வேண்டும
இநதியா என்ன கெஸ், எரி பொருள் கட்டுப்பாடா?
கவலை வேண்டாம்.
முரடன், முட்டாள் சேர்ககை / கூட்டணி எப்பொழுதும் வென்றதில்லை. தற்கால போர் மட்டுமில்லாமல் உலக வரலாற்றின் நிதர்சனமான உண்மை.
முதலாக யாரையும் எவரையும் பொது தலத்தில் கட்டிபிடிக்கிற கலாச்சாரத்தை முற்றிலும் தவிர்க்கும் படி உங்கள் ரெண்டும் கெட்டான் பிரதமர் மோதியிடம் சொல்லுங்கள். அதிலும் மத்திய கிழக்கின் இஸ்ரேயிலின் போர் வெறியன் நெதன்யாஹீ என்ற ஆபத்து அதிபதியை சொந்தம் கொண்டாடுவதை நிறுத்தவும்.
உங்கள் பிரதமர் மோதி பாகிஸ்தானிடம் தனது வெத்து வீராப்பை அட்டை கத்தி வீரனாக பிரதிபலிப்பதை அமெரிக்காவின் டிரம்பிடம், அதோடு தனது இன்னொறு பாதி இஸ்ரேயிலின் நெதன்யாஹு உயிரோடு இருந்தால், இவர்கள் இருவரிடம் ஃபோனடித்து இந்த போரை நிறுத்தவும்.
எப்படி உங்கள் பிரதமர் பாகிஸ்தானிடம் இந்தியாவின் அனு சக்தி தீபாவளி கம்பி மத்தாப்பல்ல என்ற ஜம்பத்தை காண்பித்த மாதிரி அமெரிக்கா, இஸ்ரேயிலிடம் காண்பிக்கவும்.
மற்றபடி எரி பொருள் வெறும் கெஸ், மண்ணெணைய் (Kerosene) ஆகியவை மற்றுமல்ல. மின்சாரத்தின் மூலம், உதவும் Microwave , Conventional Wall Oven, – இதில் சோறு, மற்றும் சைவமார் வத்த குழம்பிலிருந்து, உருளைக் கிழங்கு வருவல், அசைவமாரகள் கோழி பிரியாணி என்று பல உணவுகளை தயாரிக்கலாம்.
தென்னாடு – சோறு சமைப்பதற்க்கு Electric Rice Cooker மற்றும் Coffee makers, baking units and Instant Pot electric cooking unit – 1/2 மணி – 1 மணி நேரத்தில் விருந்தோம்பல் செய்ய உதவும் மிகவும் சுலபமான சாதனம். இப் பொருட்கள் ONLINE ‘லே கூட எளிதாக பல விலை ரகத்தில் கிடைக்கின்றன.
இதற்க்கு மின்சாரம் செலவாகுமே என்ற பதற்றத்தில் உடனே மூழ்க வேண்டாம். Solar, Wind, Hydro and Nuclear power மூலம், எல்லா வற்றிலும் தன்னைத் தானே ஜாம்பவான் என்று சொல்லிக் கொண்டு திரியும் உங்கள் பிரதமரிடம் இந்த பல வித மின்சார சலுகையை குடி மக்களுக்கு தரும்படி சொல்லவும்.
அதே போல் வாகனத்திற்க்கு, electric and Hybrid (fuel + electric) இந்த சமயத்தில், யார் யாரிடம் இருக்கிறதோ அவர்கள், பெரும்பாலும் உபயோகித்தால், மற்றவர் பெட்ரோல் மட்டும் உபயோகிக்கும் வண்டிகளுக்கு பயணாகும்.
Energy Saving Options: Car pool beginning with family and commuters to work – using one vehicle for allowed capacity rather than one per vehicle would save enormous fuel.
Similarly using regular public transport besides electric trains within city and areas of operation also aid in fuel conservation. Youth riding their bikes using bike lanes that is necessary in thoroughfare and busy traffic is overall good for health and natural environment.
Intelligence without common sense is nonsense.
இப்படிக்கு பொது நலம் வழங்கும்,
பத்மினி அர்ஹந்த்
Act of Benevolence is from Almighty God Not Agents of Mayhem
Act of Benevolence is from Almighty God.
People of Iran, Gaza and wherever under attack from Evil forces and collusion.
Any relief and healing is provided by none other than Almighty God.
All due credit and gratitude duly belongs to all-knowing and all-seeing Almighty God – whom you pray to and however referenced as – Allah, Kudah, Lord, Supreme Light and Being.
No entity or individual as head of the State, nation and any other are entitled to any acknowledgment, when those hogging undeserving recognition are openly engaged in fueling the fire and inflicting deaths and destruction in collusion with Evil syndicate as death merchants and contributors of carnage.
People of Iran and Palestine…when your prayers are answered by Almighty God, whom you worship, that is confirmation of God’s mercy and blessings appropriately attributing gratification to Almighty God,
Not some of those holding designations and titles as nation’s head bagging the compliment and accolade they ever starve for whether worthy or not in the superficial political theater.
The one receiving any exemption on Strait of Hormuz despite remaining hook, line and sinker with murderers of children and civilians in Iran. and aftermath in the entire Middle East are exploiters and major opportunists and do not qualify for any concessions given their deliberate misdeeds and alliance.
The same Evil clique did not hesitate to scapegoat God for their Evil action unleashing lab manufactured corona virus and profiteered from the pandemic while shamelessly propagated their Evil doing was an ‘Act of God.’
When protection and rescue received, God’s work is always behind it and in progress.
Contrarily, all negative and counterproductive activities are invariably by Evil doers and those servile to them.
Syrian State decimation and installation of al Qaeda terror outfit leader as the President of Sovereign nation Syria is the work of art of none other than the architect of tumult and turbulence – Barack Obama, whose obsession to topple the former Syrian President Bashar al Assad and handover Syria to terror group was a long aspired political dream.
Barack Obama as Dajjal / Anti-Christ sponsored terrorism and cannibalism together with Israel as well as Turkey under President Recep Tayyip Erdogan against Syria in 2011 – 2016 so-called Arab Spring is one of diverse demonic involvement by the deranged democrat Presidency of Barack Obama.
The megalomaniacal despotic regimes and agents of catastrophe claiming praise for any positive and peaceful development are nothing more than the competitors in pseudo hall of fame.
All thanks and appreciation for kindness and saving humanity goes to Almighty God and not any particular nation or the head of that nation.
Padmini Arhant
Illustration – Dajjal / Anti-Christ viz. Barry Soetoro aka Barack Obama Desperation Depiction
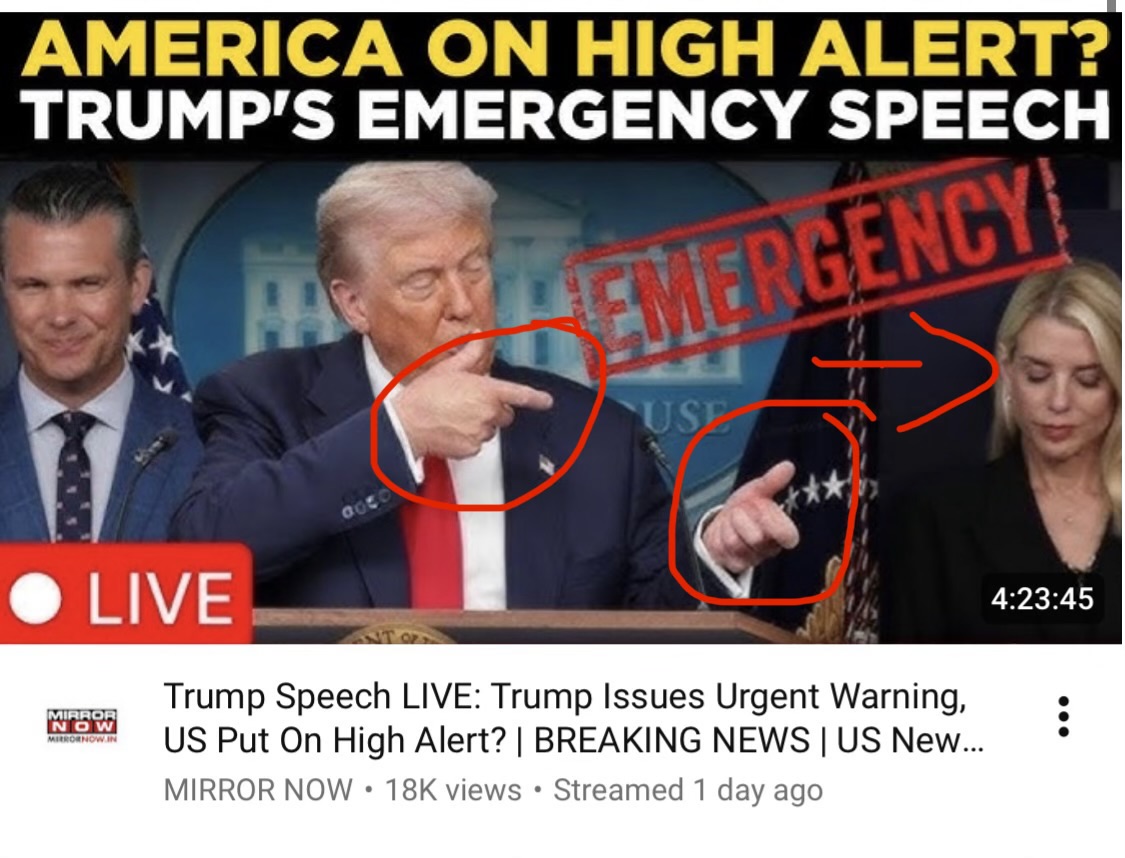
Illustration – Dajjal / Anti-Christ viz. Barry Soetoro aka Barack Obama Desperation Depiction.The obvious in this image is the remaining 9 fingers directed at the trigger puller, in accordance with the one who lives by the sword dies by the sword is applicable to Dajjal / Anti-Cbrist i.e. Barry Soetoro aka Barack Obama using Donald Trump as vector.
BTW Why is Dajjal / Anti-Christ i.e. Barry Soetoro aka Barack Obama obsessed with eliminating Pam Bondi?
The Dajjal / Anti-Christ Barry Soetoro / Barack Obama misogyny was out in the open using vector Donald Trump to fire DHS Secretary Kristi Noem.
Then wiped out female children 6-12 years of age in elementary school by Tomahawk missile strike in Iran. Now targeting Attorney General Pam Bondi.
What more?
The Anti-Christ / Dajjal viz. Barry Soetoro aka Barack Obama gone berserk, and is after the State of California in what he is adept at – false flag event made public by Dajjal / Anti-Christ without mincing words to frame Iran and hoodwink California.
Why State of California not any other State by Dajjal / Anti-Christ aka Barry Soetoro / Barack Obama?
When the end is near upon the violent assassin and fanatic killer as personal profile and political legacy, the mind has no control leading to self-destruction and mortification. That is precisely certain for Dajjal / Anti-Christ viz. Barry Soetoro aka Barack Obama operating through conjured vectors until now.
The wanton publicity drive by Dajjal / Anti-Christ viz. Barry Soetoro aka Barack Obama is sickening ad nauseam.
Among several proxies for Barry Soetoro / Barack Obama, the Trump administration VP J D Vance is another plant by Dajjal / Anti-Christ.
JD Vance profile and upbringing adapted to Barry Soetoro’s background. JD Vance drag queen get up during Thanks Giving in 2024 is one of another Barry Soetoro’s woke culture syndrome.
Similarly, Barry Soetoro / Barack Obama interjection of the democrat party member Tulsi Gabbard, the National Security Advisor, in current Donald Trump administration is the Indian name such as Tulsi and Hawaiian linkage, to establish Barry Soetoro’s Hawaiian connection imposing impulsive disorder to link with semi-Indian identity in the mega political opportunism and subversion of reality.
Not surprisingly, the failed Presidential democrat candidate Kamala Emhoff Harris introduced and installed is another product of Afro-Indo biodata for Barry Soetoro / Barack Obama to forge personal identity by proxy.
Why is Kenyan descent Barry Soetoro / Barack Obama obsessively engaged in claiming and / or latching on to Indian identity anyhow and somehow whether JD Vance with Indian wife, Tulsi Gabbard with Indian first name and Kamala Emhoff Harris with Indian name and maternal lineage to Indian community?
In other part of the world, India where Dajjal / Anti-Christ has tasked himself to creating chaos and confusion, and that too in the State of Tamil Nadu via Tamil Film Industry actor Vijay alias Joseph induction in politics and theatrics played out not without devouring lives over there as well.
There was an incident in one of the cities Karuvur (கருவூர்) where the Anti-Christ’s proxy Joseph / Vijay was vectored to hold campaign rally that ended in 40 or more civilian loss of lives in that event .
The actor Vijay instructed to make appearance well after a month of the planned incident leaving the public, and politics in Tamil Nadu to grieve over public casualty at the Dajjal / Anti-Christ orchestrated massacre.
Dajjal / Anti-Christ viz. Barry Soetoro / Barack Obama violence and quest to consume lives spread far and wide.
No matter how many proxies, puppets and pawns are manufactured representing Dajjal / Anti-Christ viz. Barry Soetoro / Barack Obama, and cohorts like India’s Narendra Modi and Israel’s Benjamin Netanyahu, they remain fraudsters in self-deception and those gullible to the charade.
God’s will is resolute and immutable regardless, with original intact in loyalty and commitment to the one and only Almighty God, and humanitarian service.
Unlike the smoke and mirror for political, economic gains and publicity mania prevalent in the impostor inventory under Masters of Duplicity.
Padmini Arhant
Attention: My Adversaries as Murderers and Evil Plotters
Attention: My Adversaries as Murderers and Evil Plotters
The fake fudged health reports you piled against me and fraudulently spread vicious propaganda on cancer, the devilish indulgence is against you with every one of you individually and collectively proven cancerous and carcinogenic in the world.
Your EVIL assassination plot against me has had the opposite effect – stronger, healthier and better than ever. 💪
Never ever mess with other’s health and life. Nor take it for granted. You reap that you sow in life.
Here goes your report and your monstrosity against me. 🤣
What you EVIL monsters in your diabolical intention tried against me is now confronting you. NO KIDDING.
Accordingly, get ready for your own grave, the one all of you desperately dug for me. All of you digging the grave entitles you to be in it now and forever.
Your grave is exclusively yours and not mine.
As for the identity thief and fraudster of Afro – Indo origin Kamala Emhoff Harris – unsubscribed email to me warning about mortality in 107 days citing cancer with taunt and torment connotation even before the concocted health reports were sent, further exemplify the devils detail plan of action overturned against them.
Hey! Identity thief Kamala Emhoff Harris – 107 days come and gone and headed towards you and your criminal league led by Dajjal / Anti-Christ.
Something all of you must worry about with day of reckoning upon all of you in entirety exempting none.
My family personal property was attacked in the loot scoot operation in February 2025, stealing all valuables and possessions by political goons organized heists and criminals, with finger prints report withheld until now after 13 months since the heinous crime was committed against us.
The court hearing was a mockery of justice system glorifying crime and made unaccountable in the democrat State of California.
The extortion of unlawful penalty on taxes paid well before deadline and financial swindling continues unabated to date by demented psycho Dajjal / Anti-Christ.
Not satisfied with the grand larceny and the charade against my family.
The attempt against my life was made yet again having pulled that twice earlier. This time via manipulation of health reports and smear campaign with many opportunists and traitors from India and even some in Pakistan joining the band wagon.
What a surprise?
These activities carried out in the aftermath of losing 2024 Presidential election by beneficiaries freeloading and fleecing on me and my family forever.
The Evil Dajjal / Anti Christ and colluders only know to kill people, assassination attempts, steal, pirate, harass and bully much to own termination.
Your show is over and you are all doomed.
Devils proposed and God disposed.👌
Padmini Arhant
P.S. எங்களுடைய சொத்து, உரிமை, பாக்கியம் இத்தியாதியை திருடுபவர்களின் நிலை – மேற்கொண்ட படி ஆவதோடு, இன்னொறு எச்சரிக்கை – சிவன் சொத்து குலம் நாசம். இதுவும் நிச்சயம்.
பத்மினி அர்ஹந்த்
Regime Change – Desperate Action Required
Regime Change – Desperately required against members in following destinations.
United States – The former democrat Presidency Barry Soetoro aka Barack Obama in parallel authoritarian role termination is urgent considering havoc and bloodshed in war ravaged legacy during and out-of-term in office.
Israel – The illegitimate Premiership of Benjamin Netanyahu evading corruption charges criminal proceedings at home. The engagement in illegal warfare from Gaza to Iran and Lebanon is entrenched in war crimes to date. The regime change here is not an option rather an immediate priority.
India – The Hindutva fundamentalist fanatic regime under Narendra Modi as the Prime Minister servile and subservient to western hegemonic goals, in the process having surrendered the North Eastern region to neighbor China’s hegemony in territorial annexations in the past two decades of Narendra Modi’s regime is treason.
Notwithstanding, the communal onslaught against muslims extensively, followed by Christians in North Eastern States Manipur, Meghalaya and adjacent states, the subdivision of Hindu caste targeting the so-called Dalits, all carried out in public lynching citing religion is a major atrocity.
The oppression of people in Kashmir is an entire separate episode under this regime. In addition, sedition is actively promoted and practiced within India and Pakistan by this regime.
Barry Soetoro aka Barack Obama, Benjamin Netanyahu and Narendra Modi are existential threat endangering lives and remain the AXIS OF EVIL.
The current President Donald J. Trump decisions and actions in continuation of warfare against Iran, and the rest of the world as a result in economic ramifications is contradiction to campaign pledge to MAGA base.
The betrayal of promise to implement peace and end wars in Gaza and Ukraine is a pivot from peace to belligerence against Iran with contagion effects in the region and the rest of the world.
Upon continuation of this trend, such egregious aggression in irreversible loss of civilian lives, infrastructure damage and the world subject to unnecessary economic misery and suffering would qualify in the above category.
President Donald Trump commitment to the American republic in eliminating income tax directed at eliminating Iran as a nation is a serious diversion posing major credibility in word of honor.
Padmini Arhant
Golden Rule in Politics
Political Hardliners – Peace and Stability within nation lies in respect for citizen’s individual rights, freedom with choice, and opportunity to progress and prosperity for all.
WEST – DO NOT IMPOSE YOUR WILL ON ANY AND THE REST. Whom you favor not necessarily the BEST. Hence, not favored by those whom you expect to accept in your quest.
Politics in general – Never fake anything. Reality is revealed similar to daylight at dawn break. The extreme left or right is never conducive.
Not too hot. Not too cold. Just right.
Everything in moderation maintains equilibrium and that is the Golden Rule.
Padmini Arhant
Attention: Devils and Danjal / Anti-Christ Sex Slaves
Attention.: Devils and Danjal / Anti-Christ Sex Slaves
Dajjal / Anti- Christ – being the notorious EVIL DEVIL you are regardless of your shenanigans and your tran partner as the EVIL DUO – refrain from involving family members who have nothing to do with your EVIL connivance and abject monstrosity.
Not all are shameless Kamala Emboff Harris salivating to the bait from you – the Evil Devil and clique. That goes for the harem as news anchors and Indian entertainment industry ever at service to satisfy yours and collective Evil Patrons’ voyeurism and orgy.
Don’t take everyone for granted – you EVIL despicable life devouring cannibal Idi Amin of America.
Dajjal / Anti- Christ STOP using proxies from your race – the likes of Ben Carlson and Ilhan Omar, failing that your alter-ego, whom you rescued from imprisonment not surprisingly sharing your trait in inflicting deaths and destruction – the Hindutva fanatic and death merchant of Gujarat – Narendra Modi.
Unlike you and your tran partner Michael Lavaughn Obama, who realized America was good only after you, the worthless and undeserving catapulted to power not without conning, scamming and exploiting me and my family to date.
”For the first time in my life time, I consider myself American- Michael Lavaughn Obama.” – Opportunist ad nauseam.
Otherwise until then you both, the EVIL DUO were cheerleaders to the pastor Jeremiah Wright at the United Church, contrary to the name promoted discord, hostility, and spewed venom against United States of America, while you both endorsed that with standing ovation in your home town Chicago.
America is not the problem. The Hannibal cannibal i.e. you and those in your league are the AXIS of EVIL.
The deep state perverts and sycophants becoming obsessed with anyone’s personal life with constant fabrication and fraudulence to fit the demonic narrative amplify the indulgers’ rotten body, retarded mind and dead Soul.
Never manipulate reality and facts and above all – The indigestible Truth for the dirty deep state radical western doctrine falsifying events from the COVID pandemic to incessant warfare against humanity.
The era of subversion and distortion proven not credible making the EVIL DEEP STATE and cohorts. the enemy of humanity, peace and progress.
Anything perverse and diabolical face imminent extinction. The Deep State is precisely at the precipice.
Losers are ever weepers and mopers driven into suicidal actions.
The Day of Reckoning is upon all of you and minions dime a dozen at God’s disposal.
Padmini Arhant
India – Under Hindutva Fundamentalist Regime
The opportunists and wannabes together with social media sleeper cells under Hindutva fundamentalist regime India, Dajjal / Anti-Christ in USA, and Israel’s payroll, are urgently required to introspect and deter from terror info war and sedition practiced by rabid Hindutva ideology led by baniya crony leadership from Gujarat.
The never letting go of sycophancy to foreign colonizers and hegemony is the fundamental tragedy of India ever remaining the most invaded nation on earth.
Padmini Arhant
Deep State Western Monstrosity -The Incontrovertible Truth
Deep State Western
MonstrosityThe Incontrovertible Truth
Padmini Arhant
The deep state western policy compulsive disorder to exert hegemonic control in the Middle East and world over is a long held tradition.
Iraq’s former President Saddam Hussein in 1990 was trapped in decoy similar to that of Russia in Ukraine war in February 2022 to date.
Ukraine war, a western ploy to drain Russia in all aspects from military, economy to general development is the bare fact. Russia was reminded to that effect via this website and video presentation for the record.
Iraqi President Saddam Hussein was lured into invasion of Kuwait in 1990 providing United States opportunity to launch attacks as planned against Iraq.
The Middle East Arab allies were not willing to coordinate with the United States hegemony fearing retaliation from Iraq.
United States administration under Senior George H.W. Bush demanded then incumbent Prime Minister Rajiv Gandhi in New Delhi, India to make New Delhi available for refueling, and facilitate U.S. belligerence against Iraq.
Indian Prime Minister Rajiv Gandhi’s polite decline was met with United States’ flexed political muscle deploying usual hardline tactics to intimidate then young Indian Prime Minister.
Indian Prime Minister Rajiv Gandhi did not agree to United States demand.
The Indian Prime Minister’s stance was appropriate.
India, as an independent nation declining passive or active participation in foreign invasion regardless of Super Power or any hierarchy was a sovereign right.
Unlike the status quo under current New Delhi regime of Narendra Modi – servile to Deep State command and diktat.
When United States hamstrung on this issue, then the Indian Prime Minister Rajiv Gandhi public statement verbatim was –
“India’s nuclear capacity could then transition to nuclear capability.” upon any force or compulsion in foreign intervention.
I distinctly remember watching this on television in the United States in 1990, with American media from CNN to other mainstream airwaves going ballistic on the Indian Prime Minister’s comeback on United States administration bully pulpit to bully others whether nations or entity all regarded a fair game.
United States and deep state western ego was bruised, and the next thing that happened was the Indian Prime Minister Rajiv Gandhi’s assassination irrefutably organized and orchestrated by the deep state.
The killing fraudulently attributed to the oppressed Tamil Tigers in Sri Lanka, and made the scapegoat in the foul play in May 1991.
The deep state evil act further embroiled Tamils in Sri Lanka with brutal ethnic cleansing under then newly elected Dajjal / Anti-Christ in 2009 as the so-called ‘Hope and Change’ only for the worst, who presided over Tamil massacre and pogrom by arming then Sri Lankan government led by autocrat Mahinda Rajapaksa, with weapons cache from Israel and New Delhi Congress government guided by the deep state to falsely avenge revenge against the fall guy Tamil Tigers and Tamils in Elam, and Jaffna.
Accordingly, the current New Delhi regime under Narendra Modi surrendered to DEEP STATE hegemonic goals is the best choice for the deep state, besides the legacy of Narendra Modi ensconced as tin pot dictatorship.
PADMINI ARHANT
Divine Rule
தமிழ்நாடு – அமெரிக்காவின் இடதுசாரி இலுமினாட்டி நடத்தும் நாடகம்.
தமிழ்நாடு – அமெரிக்காவின் இடதுசாரி இலுமினாட்டி நடத்தும் நாடகம்.
தமிழ் திரையுலக நடிகர் நடிகை வைத்து அமெரிக்காவின் இடதுசாரி இலுமினாட்டி அடிமை – செத்துப் போன பேரி ஸொத்தோரோ என்ற பராக் ஓஸாமா ஓபாமா நடத்தும் கூத்து – தன் தலையில் மண்ணை வாரிக் கொள்ளும் கேவலமான தெருக் கூத்து – தனது அழிவை உயிர் பிழைக்க முயர்ச்சி தன் பினத்திற்க்கு இறுதி சடங்காகும்.
தமிழ் திரை நடிகர் – விஜய் என்ற ஜோஸப் இந்த தெரு கூத்தில் எதையெல்லாம் எவ்வளவிற்க்கு பேரம் போசியதை தமிழ் வாக்காளர்களுக்கு கூறவும். இந்த நடிப்பிற்க்கு எவ்வளவு ஒப்பந்தம் செய்ததையும் தமிழ் மக்களிடம் சொல்லவும்.
பத்மினி அர்ஹந்த்
Propaganda Agents Representing Evil Syndicate
Propaganda Agents Representing Evil Syndicate
 Tell the World How much you are hired for?
Tell the World How much you are hired for?काली करतूत और भूत के तुम सब दूत, तुम्हारी काली हरकत पर तुम्हारा मुँह काला।
کلی کرتوت اور بھوت کے تم سب دوت ، تمہاری کالی حرکات پر تمہارا منہ کالا ۔
Padmini Arhant
Where is Israel’s Prime Minister Benjamin Netanyahu?
Where is Israel’s Prime Minister Benjamin Netanyahu?
Missing, Casualty or Fled?
No News during war time is BAD NEWS.
Padmini Arhant
BiBi – Oh! BiBi
Hey! Epstein Clients
Hey! Epstein Clients
Relevantly, as Epstein clients none of you have any reason to shame and blame anyone, when your closet has skeletons tumbling on you that you would not dare open.
The smooch, body squish, molestation, slobbery and sleazy variety are common among you perverts that you shamelessly flaunt and flirt with your types in plain sight.
Padmini Arhant
What’s up with Pakistan Team Coach and Captaincy?
What’s up with Pakistan Team Coach and Captaincy?
Ask Pakistan cricket fans, experts as former cricketers, coach, and game professionals in the second defeat in T-20 2026.
The match between Pakistan and England played in the same manner like it was against India, if not worst against England has the same defeat method adopted bowling and batting disorder that was proven a failure against India.
It appears the matches against competitive teams like India and England are pre-decided to lose against these teams, considering the team selection to begin with leaving behind Abrar Ahmed, whom the nation is crying out loud for selection and use against tough teams in T-20 2026.
Similarly dropping consistent non-performers and direct contributors in losing match against strong teams would be sensible.
The exercise overriding arbitrary decision from the team coach Mike Hesson and Captain Salman Ali Aga leading the team to qualify for semi-final and final would prove their ability to pass their individual leadership quality test.
No victory no payment to team and coach could be the turning point.
Padmini Arhant
क्या जीतना मना है किसी खास क्रिकेट मैदान में? کیا جیتنا منا ہے کسی خاص کرکٹ میدان میں ؟
क्या जीतना मना है ?
ओ भी किसी ख़ास क्रिकेट मैदान में, जो जनता के पैसे से अपने नाम अहमदाबाद, गुजरात में २०२३ बनाया गया है?
लगता तो ऐसे ही है।🧐
२०२३ उद्घाटन पर ऑस्ट्रेलिया के साथ हारे। अब २०२६ में दक्षिण अफ़्रीका के साथ हार।
क्या इस ख़ास क्रिकेट मैदान के उद्घाटन पर गो माता और उसकी बछड़ी को लाया गया जैसे भारत में, ग्रह प्रवेश में किया जाता है । आजकल वहाँ पर जो नया वास्तु रीति बना है।
अगर किया गया तो, क्या उस गौ माता और उसकी बछड़ी से यह तो नहीं कहलवाया गया की, बोल जय श्री राम, नहीं तो तुम दोनों के साथ वही व्यवहार होगा, जो हम जिसे निशाना लगाते हैं, उनके साथ होता है?
तब तो गौ माता और बछड़ी को स्वयं श्री राम वहाँ से सुरक्षित ले गए होंगे ।
जाते समय श्री राम यही कहे होंगे –
अब तो मुँह में मेरा नाम यानी राम राम और बगल में चूरी रकना छोड़ दो ।
सियापति श्री रामचंद्र की जय ।
श्री रघुपति के दस श्री हनुमान की जय।
पद्मिनी अर्हन्त
——————————————————————————————-
کیا احمدآباد، گجرات کے خاص کرکٹ میدان میں بھارت کرکٹ ٹیم مو جیتنا منا ہے؟ لگتا تو ایسے ہی ہے بھائی بہوں۔۔
کیوں کی یہ ہار پہلی دفعہ نہیں ہے۔
۲۰۲۳ میں ایسی میدان کے ا پر، بھارت آسٹریلوا کے فائنل ہارا اور اب اس وقت ساؤتھ افریقہ میب ہارا ، لگتا ہے بھارت کے ٹیم کو اس میدان سے فائدہ نہیں نقصان ہی حاصل ہوتا ہے۔
کچھ تو ہے اس میدان کے بارے میں جو جیتنا منا ہے ثابت ہوتا ہے ۔ 🧐
پدمنی ارہنت
T-20 2026 Pakistan’s Team Coach – Mike Hesson’s Contradictory Decisions
T-20 2026 Pakistan’s Team Coach – Mike Hesson’s Contradictory Decisions
The latest T-20 2026 match, Pakistan vs. New Zealand scheduled on February 21, 2026, fortunately the event was cancelled due to rain and saved by nature.
However, the valid question remains from Pakistan cricket fans, former cricketers, and experts in the field on Pakistan’s coach Mike Hesson’s controversial decisions against logic and basics, ignoring ground conditions such as pitch advantage following rain with overs reduced than pre-defined T-20 in the game.
Pakistan’s coach Mike Hesson’s decision against native New Zealand to bat after rain instead of bowling is fodder for meaningful discussion.
In this match, the former experienced bowlers, batsmen, and coaches in Pakistan unanimously agree on bowling to be better and winnable option taking weather pattern and Pakistan team’s present bowling strength into consideration.
Under these circumstances, the skepticism and disappointment against Pakistan’s coach Mike Hesson’s action upon winning toss combined with team selection as well as designation, allude to lack of relevant expertise and / or misplaced loyalty, especially in this instance the competing team being New Zealand, the home team of Pakistan’s coach Mike Hesson.
Nonetheless, the prayers from Pakistan cricket fans in the holy month of Ramzan / Ramadan is answered on this match.
Conventional wisdom – anything obviously not right fail in the fight.
Unnecessarily referencing Philosophy in anything unrelated is unphilosophical by anyone regarding themselves veteran in anything, and whomsoever they represent as monetized surrogate.
The civil and humble would not impose themselves or others on anyone they want to frame as them via emoji or expression. Only the desperate and despondent indulge in such trait.
Padmini Arhant
Satan’s Decoy and Connivance
The Satan in reference is the one that engage in extortion, exploitation, organize and orchestrate heists against private citizens and personal property to loot and scoot withholding finger prints report.
Then controls California State Democrat politics to harass, bully and terrorize target and family in perpetuity. Turns around and cries foul using Skin and identity politics in the indefensible offensive criminality.
Anyone willing to trade place as Satan’s target i.e. those in collusion with this Satan and alter-ego syndicate?
That would prove Satan’s surrogates loyalty one way or another.
When it is about others, there is absolutely no problem in collusion with Satans and beast following Satan’s script in Television morning shows, and other media propaganda outlets to taunt, mock and vilify for ratings and capitalizing personal interests.
When the sword is directed at Satan’s cronies by Satan, which happens sooner than later, the reaction and response then would be obviously different.
The appeasement of Satan while taking God’s name is typical obedience to Satan. The deceit against God by those calling themselves believers amid in adherence to Satan’s evil shenanigans are in conflict with God and truthfulness.
Such contradiction eventually cost the guilt ridden deceitful deprived of Soul emancipation.
Warning: Satan and alter-ego syndicate abusing taxpayers and electorate mandate against them as estcabilished terrorists with several assassinations and homicide in their personal and political career could manipulate and subvert their criminality to victimhood.
However, Satan and alter-ego syndicate can never hoodwink the all-knowing and all-seeing, the one and only Supreme Force – Almighty God’s Supreme Judgment beginning with dismissal of any political impunity to the Satan and alter-ego syndicate protected to date by Corrupt System on the brink of imminent collapse.
Satan’s Decoy and Connivance
People surrender themselves to the devil and the beast, and become slaves of the beast. Then Satan uses them as his weapon and makes them his own prey. Those who obey the command of Satan betray God in the name of God.
You can’t take God’s name and be obedient to Satan. That makes those servile to Satan in complicit with the beast to deceive the gullible and naive targeted with Satan’s plan to turn the tide against God.
Such collusion with devil is a grave Sin and burdens the Soul with immense suffering beyond peace and salvation.
Betrayal and treachery are devil’s signature trait and attracts the weak and vulnerable with wavering faith on God, besides mistrust on own intuition falling into the devil’s trap.
Rejecting Satan in any shape and form would demonstrate wisdom overriding impulsive behavior heeding Satan’s call and cunning strategy.
Trusting inner conscience witness to all thoughts and deeds combined with unflinching faith in God form the light dispelling darkness and reject Satans’ dark agenda.
Padmini Arhant
मान न मान हम तेरे हैवान।
मान न मान हम तेरे हैवान।
यह संदेश उनकेलिए है।
जो दूसरों की निजी ज़िंदगी में टांग अड़ाना अपना पैदायिश हक समझते हैं।
ख़ासकर दूसरे के बेटे की ब्याह को लेकर ताने मारना, जब यह भेड़िए उस सिलसिले पर कई बार दख़ल अंदाज़ी करके, जान पूछकर समस्या खड़ा करने में कोई कसर नहीं रक्के।वो एक तरफ़,
और दूसरी बात – कोसों मील दूर बैटे किसी अनजान लोगों के बारे में जिनके सात, ना कोई नाता रिश्ता था, ना है, ना कभी होगा, उनसे मूँह लगना इनका रव्वैया और पेशा है, जो अफ़सोस और शर्मनाक है।
क्योंकि ये लोग अपनी आँगन छोड़, किसी ग़ैर के निजी ज़िंदगी में दख़ल देते हैं, इसलिए इस बकवास को खत्म करने के लिए, इन्हें यह याद दिलाया जाता है, की ये पहले अपनी और गौर करें, ध्यान दे, क्यों की हमेशा किसी अजनबी लोग के बारे में अनाब शनाब बकने से पहले, सबको अपने ग़ैरेबान में झाँके देखना ज़रूरी है।
तभी इंसानियत और तहज़ीब झलकता है।
ना की अपने घर की गंदगी और नुक्स को देखी अनदेखा और इंकार करके, किसी अनजाने पर वार करना बेमतलब बेवक़ूफ़ी है ।
भारत और पाकिस्तान दोनों देश में फिलहाल बहुत औरत और मरद जिनकी उम्र ४५ – ६० के करीब है, जिनकी अबतक शादी होने की कोई नाम और निशान नहीं है।
इसलिए इन दख़ल अंदाज़ दलालों को उनकी शादी करवानी चाहिए, अगर दूसरों की शादी को लेकर इन्हें इतनी फ़िक्र है ।
और वैसे भी, यह खुली राज़ है की शादी शुदा एक हसी मज़ाक़ बन गया है इनके समाज में, जहाँ ना शादी क़ायम है, नाही वह शादी शुद्ध है, जहाँ अपने धन दौलत की नुमाईश के लिए शादी रचाया जाता है।
और फिर दहेज़ से लेकर और कई सामाजिक बातों को लेकर, शादी टूटकर रास्ते अलग अलग हो जाते है।
यह हालत भारत और पाकिस्तान में हकीकत है जो लाख चुपाने से भी नहीं चुपती, जैसे सच और मूच नहीं चिपाया जाता।
इसलिए इन मान न मान हम तेरे हैवान को यह आख़िरी बार चेतावनी है की अपने टेडे मरोड़े घर संभालें और बेवजय हमसे मूंह लगकर अपनी नाक न कटवालें।
रहा सवाल उन कच्छड़ पट्टी का जो ऐसी बातों में उलझते है, और ऐसे फ़िज़ूल बातों की जड़ हैं। अपनी भीभी छोड़े अरसे हुए, और वो भी बिन औलाद जो अपनी चिता पर आग देने के लिए भी नहीं है। इसलिए इस हालत में अपनी सोशियल मीडिया से ही किसी को भाड़े में लाने की मजबूरी है।
उन्हें किसी के जवान बेटे पर तीर मारना और ताने देना सियासी यानी राजनीति गुंडा गर्दी है। जिसकिलिए ये बदनाम है । और उसके मुताबिक इन्हें टोस जवाब लेने की आदत बना लेना ज़रूरी है।
ऐसे मान न मान हम तेरे हैवान की हरकत – अपने मुँह ख़ुद काला करना होता है ।
बतमीज़ी हद पार करने वाले अपने बतमीज़ पर लगाम दो, वरना मुँह चुपाने को जगह नहीं मिलेगी ।
पद्मिनी अरहन्त
भूत का दूत – بھوت کا ڈوٹ , Devil’s Messenger
भूत का दूत
अक़्लमंद कभी भी नकारात्मक और मानवता के विपरीत दुर व्यवहार, दुष्टता का सहयोगी और प्रवक्ता नहीं बनते।
जो लोग शैतान और हैवान के वश में हैं, वो लोग अपने आत्मा को नकारकर इन भूत के दूत बनते हैं । इसी चक्कर में यह स्वयं ज़िंदा लाश यानी अजीवित साबित होते हैं । इस स्थिति में, इस भूलोक में अपने आप को ना घर के ना घाट के पाते हैं।
पद्मिनी अरहन्त
——————————————————————————————
بھوت کا دوت
اکلمند کبھی بھی شیطان اور حیوان جو انسانیت، رحم اور انصاف کے دشمن ہوتے ہیں، انکی قابو میں آکر انکی حمایت نہیں کرتے ہیں ۔ حالاں کی اگر انمیں ہمت ہو، تو فِر انہیں ٹکراتے ہیں ، یا کمسے کام انکی آواز نہیں بنتے ۔
جو حیوان اور شیطان کے شے میں آکر انکی فطر اپناتے ہیں ، وو لوگ اپنے آتما کو حیوان کے حوالے کرتے ہیں، اس حرکت سے یہ خدا سے اپنی درمیان بڑاکر ، نا گھر کے نا گھاٹ کے ہوتے ہیں ۔ یہ اپنی مرضی سے کرتے ہیں کیوں کی انہیں عقل ہونے پر بھی ، اسکا استعمال نا کرتے ہوئے سیٹھی اپنے آپ کو شیطان اور حیوان کے دوت بن جاتے ہیں ۔ اسے انہیں بہت تکلیف اور دکھ کے علاؤہ اور نصیب نہیں ہوتا ۔
یہی حالات ہے ، ہرکوئی جو حیوان کے ہمدرد اور حکم ماننے والے ہوتے ہیں ۔
تب یہ خدا سے بہت دور چلے جاتے ہیں ۔ ۔ اس وجے سے انہیں کبھی بھی چائے اور امن نہیں ملتی ۔
اسلئے ہمیشہ اپنج کرتوت پر غور کرنا ضروری ہے ۔ کیوں کی گلٹی کرنے سے بھی ، اس گلٹی نا دہرانا اکلمندی ہے ۔
پدمنی ارہنت
——————————————————————————————-
Devils’ Messenger Impulse
The wise and discerning would never volunteer to be the Devils’ messenger and surrogate. The impulsive surrender to devil is selling one’s SOUL leaving such soulless life nothing but dead in living.
The devil epitomize chaos, confusion and carnage. Anyone treading in that path only confronted with doom and gloom leading to self-inflicted misery.
The smart and alert would deter and defy devil’s decoy and divert attention within to adhere inner conscience voice for intuitive guidance.
Better late than never saves the Soul from agony and agitation towards salvation.
Padmini Arhant
Trick or Treat – ZERO SUM GAME
Trick or Treat – U.S. – India Trade Feb 2026 Deal
Deal or No Deal – ZERO SUM GAME
$500,000,000,000 trade in return for 7% reduction in tariff from 25% to 18%.
The deal is a ZERO SUM GAME.
Padmini Arhant
Devil’s ADVOCATE -The Psycho Psychologist
When the Indian Origin so-called Psychologist is a stand by Devil’s Advocate for the psycho devil and EVIL, the clinical definition extends to all involved in psycho drama for monetary and controversial purpose. Not barring self-mortification.
Padmini Arhant
T20 2026 Cricket Tournament
ICC Politics with Inevitable Financial ICU Outcome
Padmini Arhant
The International Cricket Council taken over as Indian Cricket Council through corruption embedded BCCI – The Board of Control for Cricket in India is a case study for ICC to inevitably land in financial ICU.
BCCI renowned for incentives viz. bribes and assortment short cuts and shady deals to buy favorable decisions is classic degenerative politics prevalent in Indian political realm leading the sport cricket, originally claimed as “the gentlemen game”, drastically transformed into Indian political status quo i.e. partisanship, horse trading and gross violations.
ICC at BCCI’s behest flouting standard rules against Bangladesh ousting the team from the international T20 2026 tournament with decisions once approved against those citing security threat such as Britain against Zimbabwe followed by Australia, New Zealand, South Africa against their expressed destinations and,
India declined against Pakistan in Pakistan, despite the absence of any such possibilities with guaranteed security were refused by BCCI chaired by Indian politics.
All of the above teams from different regions’ requests were granted by ICC allowing them to exercise preference.
When Bangladesh Cricket team shared similar concern on legitimate grounds about playing the international tournament T20 2026 in India, especially with,
New Delhi politics at the helm direct involvement in the assassination of Bangladesh populist youth leader Sharif Osman Hadi on Dec 11, 2025.
The cross border state terror and murder of foreign citizens in foreign land in Bangladesh is the latest in the slew of Indian politics’ terror overseas involving assassinations of foreign nationals.
Notwithstanding, the recent Balochistan war against Pakistan army resulting in 80 Pakistan armed forces casualty and 18 army personnel held in Baloch fighters custody trace New Delhi politics considering Indian political involvement in Balochistan is an open commitment.
The ICC at BCCI coercion barring Bangladesh from the sports event T20 2026 clarify Indian political corruption presiding over international board, that is expected to be fair and non-partisan maintaining general rule across the participating nations, rather than exceptions for majority while isolation and denial against political targets within sports consortium.
The trend portends serious financial setbacks and economic liability for ICC in a sport with investments risking ICU condition under BCCI overarching political sabotage.
Any international organization such as ICC remaining independent without compromising ethics, principles and rules would not have to deal with reputation management bound by credibility factor and importantly imminent major financial collapse.
Padmini Arhant
Stupendous Stupidity
The man who calls a woman STUPID is introduced to bare facts with the reference reverted to the one of such opinion.
If a woman is Stupid, then the following beckon attention.
In english language, the opposite gender to male is referred to as – Fe(male), Wo(man), and the pronoun S(he).
In all of these references, the male, man, and he follow the prefix or the first letter.
If a woman is Stupid, then what makes the one following her in factual reference? If not stupendous stupid.
In other connotations – Mot(her), Fat(her) and Brot(her) – have her presence and relevance in relation and affiliation making the terms inseparable without ‘her’.
Something to think about for the ones who think they are genius and the woman is stupid. 🧐
Padmini Arhant
Signs of Defeat
Dispelling the myth on “Woman has no friend” says the ones surrounded by the deceitful, baneful and woeful wary and fearful of the scary dagger drawn against them from within, the so-called friends, is a treachery in disguise as friendship.
FYI – The Woman with no friend cried out loud by the jungle chorus fail to recognize the fact, that she is a fortunate and blessed Soul to have the Supreme Light as the Soul Mate in essence and presence beyond time and spacial limits.
The prominent signs of defeat.
1. Denial of reality.
2. Rattled and perturbed at mirror reflection.
3. Gather proxies and pawns to defend the indefensible status ending in exacerbation and expulsion.
4. Nothing to live for except fighting the lost battle for the nth time inviting embarrassment and disgrace.
5. Attention sought is attention denied in fait accompli.
Padmini Arhant
Brilliance, Magnificence and Fragrance
Anything natural and original requires no publicity and propaganda or imposition in perpetuity extending beyond long expired term and time.
The brilliant Sun needs no introduction nor staged orchestration of its presence.
Similarly, the pearl white moon shines with magnificence in defiance of the pitch black nightfall with transitional darkness limited from sunset to sunrise.
Both Sun and the Moon by natural design, permeate light with Sun dispelling darkness, and Moon light together with multitude white diamond like sparkling stars host the Sky and earth with dazzling radiance amidst darkness at the beginning to the end.
Light dispels darkness and not the other way around is profound in nature’s tapestry.
Likewise, the sweet fragrance of a flower draws the sensitive towards the direction without the flower desperately seeking attraction.
The stark distinction between real i.e. natural and the one in direct contradiction are visibly exemplified in characteristics.
Padmini Arhant
Self-Abandonment
Lack of Self-esteem is Lifeless i.e. declaration of demise.
1. Those who run away from own identity and profile to be someone they regard lesser of them. Not realizing the foolish indulgence amplifying self-degradation.
2. Imposing themselves on the one they envy and despise the most is offloading them in the obvious status unable to tolerate and accept as to who they are in reality.
3. When you have no respect for you desperately wandering to be someone except you, how do you expect respect from others?
4. Available to be used as pawn for all things negative and indignation is confession on own character beneath value, and devoid of honor. That is also acknowledgment of self-disgust prompting self-abandonment to be the other than them.
5. Those having problem in accepting their identity for whatever it is worth, and instead obsessed with anything that is not even remotely associated or related is a serious personality disorder.
Above all, DNA manipulation is demented deploy futile against the indomitable nature behind all creations.
Padmini Arhant
Politics’ Fake Guarantee
In politics fake goes a long way than facts. The latter is considered inconvenient and meddlesome in politics.
The politics that claims to be the savior, defender and protector of the people and country would not dispatch next of kins – son and daughter away from such secure homeland to a foreign land especially national security under personal control.
The politics that cannot guarantee own family safety prompting safe passage of them away from the supposedly solid rock home turf.
Meanwhile expects citizens in the country to rely on the political leadership, who is not personally related to all members in society to trust their life under such stewardship.
Anything goes in politics to stay in power is the fact. The reality is never reconciled with in the power greed manifested dark age.
The political irony unfortunately is a burden and liability for the people and nation to risk their life under such lifetime occupation declining to cede power unto death do apart.
Padmini Arhant
Crime Evasion by Compulsive Criminal Disorder
Crime committed since adolescence to adulthood extended lifetime in sync with cradle to graveyard is a compulsive criminal disorder always finding illegitimate excuses to evade indefensible criminality lifelong.
The devil in disguise could no longer prolong cloak and dagger profile.
When committing brazen crimes such as summary execution of a rape victim from own race and ethnicity viz. African American, deserting own child from the rape seated in the back seat with full knowledge, and still targeted with firearms via tax payer funded security in broad daylight captured in world view on camera never held accountable.
Despite the murder near the periphery of taxpayer funded public house, the White House at 1600 Pennsylvania Ave, Washington D.C. United States.
The reason behind letting the authorizers responsible for the savage killing in 2014 off the hook then, as sitting President and the First Lady involved in murder of own citizen with malicious objective to get rid of own child from the rape cited as political impunity.
During and post term in office, the criminality to harass, bully, violate and commit violence against targets in civil society is self-granted prerogative.
The ongoing criminality having gotten away with such transgressions during the official term in office holding self, the next of kin, and those involved in the audacious public execution of members from own community, and any other like the disabled man on wheel chair victimized in sodomy and eliminated thereafter, the murder of the chef at own residence…among several known and hidden criminality is the cumulative violent saga treated as Presidential privilege.
When the past persists at present to be continued in the future based on well substantiated trajectory is conveniently argued as old events brought to surface.
Notwithstanding, the crime never desist with criminals abusing political status remain Scott free taking other’s life and rights for granted forever, and continuing with business as usual justifying mob murderous indulgence as political entitlement.
The proof is in the pudding when the guilty desperately and infinitely running away from self and own reflection unable to reckon with personal reality and unforgivable crimes in the criminal political culture.
When you are not at peace in living and perpetually engaged in anchored sabotage and disruptions of those envied with overwhelming malice, your conscience is witness to your flagrant inescapable crimes and guilt ridden existence.
Such living is a punishment from own karmic woes and debts never settled instead burdened on Soul with no salvation.
Padmini Arhant
Female Impostors Clad in White
The recruits of EVIL as social media anchor and a plant in government ironically for security, clad in white while deeds are dense black as impostors are VIXENS guarding the chickens den.
Those who fool others are the first to be deceived by own deceit.
Running away from self to be someone else is a personality complex with little or no self-esteem.
The one who is afraid to be oneself is self-convicted reality.
Padmini Arhant
The Unprecedented Prejudice – The Western Fatwa
The Unprecedented Prejudice
Padmini Arhant
To: The she-bashers and misogynists from applicable destinations and affiliations.
If you claim the content and material published thus far on this site and sub-domain in the past nearly two decades, originating from high above the Sky, prompting you to set up pseudo platforms as Facebook, followed by Twitter and later the Sky News Australia – drawing from my past life residency in that domain, then you need to pay attention to the message below.
Although, these social media and Sky News mainstream media are the political syndicate’s propaganda channels to counteract the real substance presented on this website and sub-domain.
Then those, for whom I am the anchored target, individually and collectively need to take your contentions to the one up above in the High Sky, rather than spewing venom and vitriol with assassination attempts against me including manipulation of my health records with malicious intent, besides my financial accounts and more, alongside the criminal heist orchestrated against my Apolitical family, who have absolutely nothing to do with politics.
The criminal loot organized against my family upon losing Presidential election in 2024 is also subverted despite holding the finger prints reports months after the vicious crime stealing the personal documents from birth certificates to passport etc.
Not to mention making a mockery of court hearings on this open shut case, with politics controlled judiciary, all of whom funded by our tax dollars exploited against us.
Then with that knowledge having been directly involved in the crime stealing documents, the democrat political rule in the State of California is misused intimidating the victim to submit the original document to accept the filed personal tax return with due tax refund denied from the State of California.
The condemnable sleazy politics and lawlessness against tax payers and law abiding citizens are exercised as the political might.
What more?
The ludicrous phonetics ending in ni like in Bani, my as in Jimmy, ny in Clooney…linked to my first name Padmini – Pad(mi)(ni) are all dumped on me in a bizarre resignation of anything goes to violate the rights of my family and me in the last two decades in the name of western democracy.
This is the devil’s artwork protected by evil clique with a green signal on satanic activities against me and my family.
London with the deceased monarchy unrelenting to cede throne to own heir next in line until demise had no problem on Royal decree for anyone to be assigned with my identity in a manner of auctioning my life at these narcissists as self-proclaimed elites’ discretion.
The pertinent question that arise on the opportunism granted at my expense to all those predominantly in politics, entertainment industry, media, beauty pageants, and even religion is, all of them have been around going back to 70’s, 80’s and 90’s long before my public appearance, though I have been prohibited from engagement since day one to date on private media and public channels in blatant discriminatory practice against me.
How come it never occurred to them then on divinity and angelical aura they are claiming to possess now at external influence and nominations after my public entry?
The agenda until now has been to deprive me from any fair audience interaction fearing threat to their seized opportunity and wannabe moment.
Interestingly, them pretending to be me is the X factor experiment to the extreme proven unfavorable for many on the beaten track.
The darkness is dispelled at dawn break and cannot be extended over against natural settings.
The built-in hideous acrimony from those having illegitimately attained anything in life surface upon finding anything diametrically opposite to them in the truth vs. false.
London of all the places neither have credibility nor justification to pass judgment on my inalienable right to oppose my individual rights violations that are selective and subjective for them rather than objective and inclusive.
Accordingly, make up your mind.
In your arrogant opinion, if I do not have the brain and intelligence to function and perform any tasks, unlike you and the ones in collusion, then refrain from embarrassing political abuse of power and authority against me and my family as your given right.
You can’t have it both ways.
Regardless, no State or non-State elements and factions have any right to intrude in anyone’s personal life, home and existence that are exclusive and private, similar to the intruders assuming their rights in all these aspects, and yet create havoc against the target as their entitlement.
The lewd perverse conduct is shameful and reprehensible.
The goon hooligan politics and the crony contingency from the Indian entertainment industry and media elsewhere stooping to any level in the constant vilification and psychological perversions wishing and hoping for my disappearance, sooner than later to be experienced by every one of them, permanently ceasing the charade is the only option for the howling hyenas in the otherwise futile confrontation.
Upon speaking truth and factual presentation on political status quo and any matter, the constant virulent she-bashing maintained by political misogyny is by far the obtuse intellectual deformity.
Intellect and intelligence is presumed male character and exclusive proprietary heritage displaying narrow mind and tunnel vision in the process, not necessarily attractive for the source.
The political dementia taking 180 degrees turn prominent upon non-compliance of objectionable actions.
Before election to the highest office in 2008 Presidential race.
In October 2024.
The disparagement unleashed beyond civility and decorum by the one as the Nobel Peace Laureate and Presidential status upon declining unruly mob politics’ coup dètat and myriad violations in the Presidential tenure.
”Oh! You not only have the booties, you think you also have a brain”?
The obscenity ad nauseam from the one crying foul on racism, civil rights, liberty and justice…obviously only reserved for them.
The political decadence expect those to whom they are hostile to accept the culture of insult and insolence, invasion of life, space and all things personal and private, the gross violations as an experience to be cherished not chastised in their twisted interpretation.
Reiterating earlier passage on this site on Universal female significance beginning with;
Mother Nature, Mother Earth, Mother Land, Mother Tongue…are the fundamental epitome of pervasive female orientation transcending race, religion, and diversity since world creation continued to the world without end.
The female X chromosome in genetics also represented in the XY male identity by natural design. The deletion or omission of X chromosome at will by misogynists openly and violently to purge female existence would only purge them, for the female identity is independent in XX representation.
Whereas, the male Y chromosome denoted together with female X chromosome contribute to male XY factor in the male identity – in the birth of a male child.
That aside – in English language – the pronoun symbolism that was invoked predominantly to infuse the confused trans mania under democrat Presidency from 2009 – 2016, unfortunately reemerged in 2021 – 2024 and extended to date,
The term fe(male), S(he), Wo(man) – identifying the opposite gender of male, i.e. female is self-explanatory in the word order on the prefix and suffix.
The political pejorative mindset subjecting women as a fair game deteriorated since 2008 for the worst, and thereafter downhill is not limited to the BJP coalition partner, the Indian politician, the Chief Minister of Bihar, Nitish Kumar pulling the hijab from the face of a woman of different faith in public to body shaming in optics as the norm using women from entertainment industry with the focus on their horizontal expansion and worse…
Among these many aimed at the obsessed target – the arch enemy, i.e. me, the woman whose name is withheld from public reference, despite the name right in their face via presentations in writing, visual and audio content in the past twenty years to date.
The despicable decree against me originating from none other than the western bastion positioned as the paradigm of human rights, women’s rights, democracy, fairness and equality…exemplified paradoxically in context.
Similar to Fatwa, the typical reactionary course in paranoia that is overwhelmingly criticized by the west upon such decree from the non-western frontier against their protected members as author in the western preferential deriding of the religion or societal defects of other cultures, nonetheless pledged allegiance to them and their colonial mentality.
While the selective human right violation noise reeks the political colosseum, varying from the London based Amnesty International and alike in sheer western hypocrisy,
My personal objection to the stampede in violation and abuse of my individual right by those as their prerogative is not even tolerated by the abusers and those complicit as silent spectators including the so-called human rights watch groups in the prejudiced predator proclivity.
Furthermore, the targeted malicious misogyny emanating from the individual misinterpretation of Homosapien representing the human race, contoured to homogenous male gender classification against the natural heterogenous origin and existence is isolated tribal attitude, appropriately befitting the crude tribal characteristic with the slang “the lone wolf” – the absurd and ironic reference used against me by the actual representative.
It takes one to subject anyone as such in denial of mirror reflection and deranged disorder.
Like everything else, the term legal on Fatwa is relative in the present time to suit those presiding over definitions and distortions. No surprise whatsoever.
Nonetheless, the ultimate decider of fate and destiny is not them, but Almighty God – the decider for all, including them delivering their verdict against anyone not toeing the line with them.
All the more reason for the World Without Politics to restore and revive sanity, decency, human respect and values bidding farewell to prevalent uncivilized, uncouth, unbridled brute politics.
Padmini Arhant
Expression of Gratitude
Happy Deepavali / Diwali! October 20, 2025
Duplicity Feigning Authenticity – Author & Presenter Padmini Arhant
The Counterproductive Political Trend (Synopsis)- English
Indian Political Intervention in Pakistan (Hindi / Urdu)
The Ordinary as the Extraordinary. आम लोग की खासियत
KBC Ko Civility (सभ्यता) का ABC में Shunya (ZERO) – in Hindi
எதிர்மறை அரசியல் திரை நட்சத்திர தற்கொலை
दुश्मनी, विरोधाबास जीवन। घर की मुर्गी दाल बराबर।
Teleprompter की महिमा (Devnagri Hindi)
அரசியலின் வெத்து வீராப்பு
அரசியல் சினிமா ஒன்றையொன்று சார்ந்துள்ளது
இளைஞர் தலைமுறை தற்காலம் எதிர்காலம்
அரசியலின் துரோகம் தீவிரவாதம்
வேதாளம் அவதி
அரசியல் திரையுலகம் காந்த ஈர்ப்பு
அரசியல் விளக்கம்
அநாகரீக கலாச்சாரம்
இனம் அல்ல குணம், நிறமல்ல அறம்தான் முக்கியம்
அரசியல் மிரட்டல் (அரசியலக்கு உறுதியான இறுதி சடங்காகும்)
அரசியலின் ஞாபக சக்தி மறைதல் (Politics’ Dementia)
அகத்தின் அழகு முகத்தில் தெரியும்
அரசியலின் அகம்பாவம் அடிமுட்டாள்தனம் அழிவின் அடையாளம்
அரசியல் விபச்சாரம்
कुर्सी की अपमान
कुर्सी की सम्मान कौन जिम्मेवार?
அரசியல் நாடகம் ஜெயலலிதா மரணம் உதாரணம் அரசியல்-நாடகம்-ஜெயலலிதா-மரணம்-உதாரணம்.m4a
ப்ரமணின் முடிவுதான் வெல்லும்
கிழக்கு ஆபிரிக்கா கென்யாவின் வேதாளம்
என் பரம எதிரி மற்றும் விரோதிகள் வாழும் வாழ்க்கை
இப்படிககு
பத்மினி அர்ஹந்த்
Natural Design
It is good to see that after the Wild Fire, the region looks mossy and lush.
Nature triggered controlled wild fire is meant to get rid of the dead bark and trees that are standing tall but typically they are dead taking the space on earth that are best replaced with fertile, green and lush growth releasing oxygen and nourishing life. It is a natural process by nature’s design.
Padmini Arhant
[memberonly] Nature – The Most Powerful Ever
Nature as the Supernatural force and phenomenon, presided and governed by the Almighty God in Spirituality is the one and only most powerful ever.
The titles in power, fame and fortune as “the most powerful person in the world” is build-up and excess adulation of the public service position and alike in the economic sector.
The occupations essentially legitimized and funded by ordinary folks across the spectrum. The people without whom, neither and none could function nor benefit from the designation.
Above all, life is fickle and the status of any kind melts down eventually and converge with the sea called the general society.
Humility and modesty are rare. Yet a necessity in elucidating human character.
Padmini Arhant
Empirical Facts
Light dispels darkness. Not the other way around.
Provocateurs and Saboteurs succumb to Self-Destruction.
Soul burdened with Sins makes life of those a deadweight on them.
Life shackled in guilt is the karmic effect unable to runaway from self, and ends in the vortex of shame and sorrow.
The verdict from within – the inner conscience witness to mind, body and spirit engagement in living determines inner peace.
Padmini Arhant
Politics – Abusing Power and Taxpayer Money
Tran Politics’ Trans-Fixation
What happens when the obvious is more transparent as the tran polity pitch reach a crescendo?
Michelle is Michael in French. The maiden name Michelle Lavaughn is Michael Lavaughn Obama.
That being the incontrovertible fact and reality, the following is apt for the tran polity obsessed with tran mania.
Hey! “You are that you think I am. I am not that you think I am.”
Your RAINBOW PRIDE is the impetus to your imbecile demeanor driven into the bottomless pit.
One can run as far as one intends, but can never hide. Cease and desist your provocative incendiary interventions in your target’s life.
Parasites declined parasitic culture and existence is naturally distraught and desperate for host.
The devil in disguise gets caught with the foot in the mouth in own exposé.
The one digging grave for others find themselves in the dug grave in the DIY gimmick.
By the way, racism against black America or Africa is coined with different connotations, the latest being “KAREN”.
Similarly, should black racism and hostility against their obsessed target be aptly referenced “Mufaso or Mombasa?”
Padmini Arhant
Traditions from Abroad – Men in Tradition

No Life Become the Low Life
Those with NO LIFE invariably become the LOW LIFE prompting invasion, intrusion and intervention in OTHER’S LIFE. Accordingly, such life succumb to demise.
Padmini Arhant
Feet, Feat and Fate
Make a mark with your feet. Not by seizing another’s feat and fate.
✌️🙌✊🧢🏀 etc., including Chicago Sky and Hawaii ocean none of these would mean the identity swap as implied and alluded in the bizarre crooked wicked trend by politics and blind following by crony contingency.
Food for Thought – Originality demands ingenuity and together defines integrity and credibility in individual character and actions.
Criminality is the trait of the traitor born incorrigibly corrupt and criminal roaming as the devil in disguise entrenched in misogyny.
Anything imposed in contradiction to reality is the bare fact on such thing being the reject and desperately seeking meritless recognition.
Juxtaposed, the bright sunshine is pervasively bright enough to dispel darkness without any harbinger to make aware of the brightness.
Yet another parody is the self-inflicted tragedy.
The attire satire – in the staged satire – the contrived White House melodrama referenced in the article below – is illuminated with irrefutable TRUTH.
The attire significance in keeping up with appearance syndrome in politics has splashed disdain all over itself, smudging and smearing the suited and booted in designer labels and alligator shoes albeit at the taxpayers’ expense.
The latter not necessarily suited and booted like the beneficiaries as it is not required like so, to gloss over the decayed rotten interior overshadowing the exterior.
However, the ungrateful and uncouth ever basking on the besmirched eternal charity is the embarrassing reality.
For the pompous ignoramus information – The white collar crimes albeit suited and booted in expensive getups engaged in grand larceny of nation’s treasury to scamming, conning and cheating, committing treason against the ones funding their existence unto their demise are by far the most dangerous and perennial threat to society and humanity at large.
These crimes continue to cause economic tumult and devastations, destructive political perversions and systemic corruption criminal culture in the core abuse of power.
All these crimes are committed in polished boots and immaculate suits blinding the abused taxpayers and hard working citizens across the spectrum in society.
The worst, they remain at large maintaining further criminal lifestyle and loot carried out with authority, while drowning in disrespectful deluge upon their behavior stripping them of the false savior hood in
Who is who? – public display.
Notwithstanding, the ones who were denigrated and described “unfit” to engage with the suited and booted diatribe posed as the dialogue in ambush eventually prevailed in history.
The non-violent pioneer of Independence movement against the brutal imperialistic power, none other than Mahatma Gandhi was ridiculed by the imperial servile Winston Churchill as – the “Half-Naked Fakir”.
Did the so-called half-naked-fakir not prevail over the booted imperial power, renowned for the loot ever?
How about the King Siddharth born with Royal insignia, renounced the regalia and adorned in simple robe, became the ascetic who enlightened humanity?
Henceforth, revered as Buddha accordingly by the ones who recognize the enlightened Soul?
That is the stark distinction between the real undiminished light and the fraud plunged in darkness.
The battle between Evil and goodness is that of devil and deus. Succinctly, the evil succumbs to vanity and unsustainable criminality.
Padmini Arhant
Western Characterization of Masculinity
Telling a joke, being competitive and drinking beer – all exclusive to male? Since when?
Aren’t there women comedians and standup-comics?
At least in other cultures, they are thriving in entertainment. Some are quite famous. The same goes in families, there are women with great sense of humor.
In competitiveness – Don’t western sports profiteer from women aggressively competing with rival teams?
Like the prominent WNBA that is not yet titled ANBA (Androgynous) or HNBA (Hermaphroditic) or,
For that matter, the bash and smash sports Tennis – U.S. Open, Australian Open Wimbledon, French Open ending in Grand Slam as the name suggests slam the series with mega bucks made by all those sharing the fortune.
Last but not the least – Beer chugging by white women is a common sight during Oktober fest in Germany and alike in Australia, Britain, Ireland and Nordic countries by white girls and women, who have beer with their male and / or female companions in western hangouts at the pubs and bar that are fairly normal in western culture.
What about western women, some high profile female political members in the United States political party, their widely known passion for Russian Vodka, ironically promoting the economy of a nation, reserved by them, an adversary.
The idea of exclusivity from behavior to lifestyle, intelligence to bravado delineated gender specific in western politics, not necessarily in western general society is a momentary relief.
The former distinction considering politics dwell in political universe that revolves around own political ideology and perception of life as linear and Asymmetric.
Above all, the presence of both male and female hormones viz. testosterone, the male hormone and estrogen, the female hormone are prevalent at varying levels in all male and female species.
The humans in particular, revealed in anatomy and physiological characteristics such as the chest in a male similar to that of a female with the latter only enlarged due to mammary glands.
Likewise, some women tend to have dense body hair, thick growth on their arms and legs from the higher level of male hormone triggering these physical features that are otherwise common in men.
In a way, the male and female composition complement one another, designed by nature in the nucleus sharing the DNA of both male and female chromosomes.
Politics might have entirely a different perspective and interpretation especially in contemporary age to fit the political criteria on anything and everything that are uniquely natural and normal.
All the more reason, for individuals from all walks of life to exercise own intelligence and discernment in making informed decision rather than falling for political narratives driven to mislead and cause confusion.
Padmini Arhant
EVIl invites PERIL
Words of Wisdom. Works All the Time 🤗
Friction or Traction. Private affairs are best kept private rather than fantasized as public.
Only those who have no life would be obsessed with other’s life. Such existence is being dead in living.
Life is a blessing not meant to be destructive to self and others.
Padmini Arhant
Tesla Inc. Unconventional Solar Roof Contract
Does anyone know the future with certainty?
If they did, they would be the ruler of own destiny.
However, the multi-billionaire Elon Musk owned and run Trillion $$$ Tesla Inc. binds homeowners on contract related to Solar panels and Solar roof in a rather unconventional practice.
The homeowners are drawn into a Solar lease arrangement on long-term basis based on the product longevity guesstimated by the company as twenty years, even though the real experience is far below well before half-way down the contract.
Nonetheless, the contract is established as zero downpayment with fixed monthly charge to bank account throughout twenty-year-contract, with additional fees upon using any other payment options.
The homeowners decision to terminate the contract at any point in time prior to contract expiry for assortment valid reasons is persona non grata – simply unacceptable predominantly clear in such outcome.
The product and service not meeting minimal requirements in Home Energy Efficiency and,
As a result, the investment proven a liability being the common factors face steep financial monetization by the company citing own policy.
The company requirement from the homeowners to pay for the remaining period of the contract such as 10 or 15 years – $13,000 to $18,000 in that 20 years lease agreement upon early lease termination is a phenomenal extract.
This is despite, the products and services are no longer used and duly returned to the company in the relevant time and condition.
How does such disproportionate deal is fair to homeowners?
The company has to be paid in lump sum upon homeowners exit prior to lease expiration besides returning the products and systems back to the company at that time.
Applying the analogy on home rental lease,
Could property owners demand entire lease term payment in three to five years lease from tenants wanting to move out or discontinue rental based on legitimate reasons?
The laws strangling the property owners leasing apartments and homes are extraordinary compared to Trillion $$$ tech giants and alike oligarchy basically owning governments and legislations in the direct buy-out with few million $$$ in political campaign and Presidential candidacy, raking fortune in return.
In the process, trading people’s rights as consumers and homeowners as applicable.
Seeking full settlement on the unused lease term on Solar roof or panels, is demanding homeowners to accept future set in stones with events writ large in life in the reality of even weather forecasts not always necessarily accurate.
Above all, generally politics and economic sector default on their respective pledge to public.
Such lopsided agreement entirely benefitting the company is a trap with the exit strategy for homeowners to free themselves from the expensive inconvenient setting stipulated to paying ransom to the company.
Apparently, marriage divorce on irreconcilable differences are lot easier and not so compelling than the divorce from exploitative opportunist politics and influential economic greed.
Regardless, Is this the pathway to Super rich MBT – multi- millionaire, billionaire, trillionaire status?
Exploitation and extortion as the means to self-enrichment and political empowerment.
Padmini Arhant
Ukraine not Ukraine’s President Seems to be the Problem?
Ukraine’s President with the last name Zelensky identifying the faith, whether religious, agnostic or atheist, perhaps would not be subject to recent nomenclature, had the President been representing Israel.
Ukraine’s President would most likely and almost certainly enjoy similar status that of counterpart in Israel in the parallel warfare in Ukraine and Gaza.
Ukraine appears to be the problem more than Ukraine President, for anyone targeting Ukraine.
The western members rallying behind Ukraine contrarily joined forces with Israel in Gaza Genocide. The western determination of invasion is the one that fits own definition.
Padmini Arhant
Scoundrel Kavodhi Funeral
Politics and Media Fanaticism
The nuanced insolence against me and my family – verbose, visual and preposterous enactments by far remain the dumbest idiocy practiced by those claiming to be the,
“Be all and End all in the World.”
There is no doubt, they are being who they are in exemplifying insanity and profanity. Nevertheless, the end of anything insane and idly mundane relevantly applicable to them is beyond their comprehension.
I understand, those engaged in this lunacy have no life. Otherwise, neither would be suffering from the Obsessive Compulsive Disorder with incessant obsession with me and my family regardless of them skidding in the process to the bottomless pit.
The allusion with anything ending in monosyllable phonetics – ni, mi, me, ne, ney mey…and for that matter the letter P – interestingly unbeknown to the OCD, also represents the Planet, People etc., on Earth.
The phonetics fanatics are the biggest joke🤣 in the outrageous sickness proving themselves non-salvageable toxic waste in the environment.
Padmini Arhant
Devil in Disguise
The Devil in disguise is no different from the Pervert with nothing on.
Those who use other’s positive karma for own promotion and narcissistic self-interest are the same as sinking boat with dead weight.
Enactments of fake situations to console the Satans is exacerbating satanic evisceration.
The villains, demons and Satans imminent end is ominous, when they indulge in rage and devilish activity.
The destruction motive against the target is hallucination yielding self-extinction.
These aptly apply to devils and EVIL syndicate perpetually fixated on their target. The obsession, inevitably expunge the devil and Evil in accordance with natural law of Karma is the done deal.
Padmini Arhant
Black Deed and Slavery
The black deed blackens face and chains the mind to slavery servile to the devil within.
The successive defeat and rejection, encountered as the ill-fate of black politics throughout since emergence to date, is black tar on the parasitic culture fleecing and freeloading on their target much to individual and collective demolition.
The pitch black dark night is devil’s comfort zone. However, the experience is short lived at dawn break and sun rise dispelling blackness to oblivion.
Padmini Arhant
Piracy Plagiarism Plague
Rhetoric vs. Reality – Piracy
Wig vs. Real Hair – Factual
Crooked (Crocket) vs. Honest – Actual
Fake vs. Original – Absolute
Sham vs. Sincere – Truth
Padmini Arhant
FREE PALESTINE – Not Arm Israel with $1Billion Weaponry
FREE PALESTINE – Not Arm Israel with $1Billion Weaponry. Padmini Arhant
Poison Politics – Death Wish
WHY DARKNESS SHUNS LIGHT?
What the contemporary poison politics should have in their Death Wish are the following;
The death of abject corruption, violent criminality, political abuse of power, taxpayers’ hard earned money, serious violations, invasion of life and rights, identity theft, piracy, plagiarism, prejudice, propaganda, hatred, misogyny ad hominem and,
Above all, the burial and cremation of political impunity and sanctuary on all these abominable atrocities and abhorrent legacy.
Padmini Arhant
Comedians as Political Surrogate
The so-called comedians in the lack luster late night shows 🥱 on multi-million $$$ price tag with pre-recorded phony applause played to their sound-bites at “the expired political term designee’s behest” is satire at own exhibit. The presenter’s performance to the empty seats, is gala comedy routine.
The prying, preying, spying, snooping and ever intrusive, invading people’s lives gutter politics entrenched in *sexual connotation* of anything, prompted by own dysfunctional personality in oedipus complex is beyond remedy.
The degenerative mind and sadistic impulse expedite individual and collective succumbing to obsessive compulsive disorder.
Padmini Arhant
Life is a Gift
Life is a gift. It is meant to be positively purposeful not negatively wasteful.
The world creation is heterogenous not homogenous. Accordingly, not all things are derived similarly in source and practicality.
None have the right to define, determine and validate anyone’s identity and intelligence or the lack thereof.
Human status is a blessing not to be squandered in idle fixation on things and persons not even remotely connected in any format.
Instead, internalization is the place to begin with to recognize the cause of such mental and emotional turbulence. Failing that professional help might be a way to sort oneself now and moving forward.
Can’t do any good. Never mind. The least is to refrain from doing any harm to self and others.
Padmini Arhant
Life – Interpretation
Is embryo to be treated like people? asks Arwa Mahdawi of the London based “The Guardian.”
People – A person’s body has countless cells that are required to be alive and well in order for life to exist in general.
For example – The aging of cells contributes to weakening of tissues, nerves, veins and arteries that in return impact the vital organs, the nervous system and the overall well being.
Embryo upon fertilization of egg from the female ovum by the spermatozoa from the male, thereupon the development leads to fetus formation in the uterus. The function is possible only when the cells are healthy and alive from conception until live birth and then onwards expanded into the life span.
As for the sex determination of the fetus – Yes it is female XX at origination until twelve weeks, with the Y chromosome from the male sperm were to be dominant altering the XX to XY for necessary reproductive organs such as male genitalia.
The discarded embryo used in the embryonic stem cell transplant saves millions of lives, cures many life threatening diseases and contributes to renewal and rejuvenation of body cells enabling better quality of life for those suffering from crippling diseases and health problems. Only living organisms and cells can facilitate life and functionality.
Embryo is a living body of cells that otherwise would not be possible in becoming the fully developed fetus and eventual live birth upon delivery from the womb.
Hey! The above is No AI B.S. or Plagiarism from Internet source.
It is straight from natural endowment – the organic brain with active brain cells in my body enabling thinking ability besides exercising reasoning faculty from within and recounting experience from two live births in my present lifetime.
Padmini Arhant
Blatant Coup détat with no Democrat Primary in July 2024
The devil can no longer be in disguise.
The one pretending to be the promoter of women to break the glass ceiling would not have been primarily preoccupied in systematically and brutally removing women leaderships as the heads of state especially in Asia, Australia and Britain sparing Africa in the particular terms 2009 – 2016.
Similarly, without a shadow of doubt since 2009 – 2016, via the alter ego in Indian politics, the trend involved in humiliating then female leadership in the State of Tamil Nadu, the late Chief Minister Jayalalitha. Not barring, the deceased CM’s death in Dec 2016, shrouded with many unanswered questions to date.
In real sense, apart from common sense, the former Tamil Nadu CM Jayalalitha was far more capable than the BO and MO recruit -B.S. – the former VP and Presidential candidate Kamala Harris.
What does that say? There is no tolerance for competence and courage in female leaderships to pushback male supremacy in global diktat presided politics.
So much for caring to see a woman with Indian descent or lineage to shatter the glass ceiling?
That too, How was that hatched by the BO and MO – B.S. duo to prolong puppet woke DEI regime?
In a blatant coup d’état against own party incumbent Presidency brazenly subverting democracy slighting the democratic primary, with the induction of failed flawed candidacy, whose track record reeked dismal performance, for which the candidate, Kamala Harris was long rejected in the 2019 Presidential bid.
The reality in the candidacy was not Indian or Indianhood rather AFROCENTRIC – to reflect own BLACK race prevalence, with the candidacy touted as the so-called ‘the First Black Woman’.
Otherwise, the 2024 Presidential democrat candidate could have been from open field democratic primary by election *not* selection. Accordingly, the democratic process was rejected for obvious reasons.
Even with the choice of Kamala Harris, what was the exhibit?
The former VP Kamala Harris naturally and voluntarily in adherence to the script from the puppeteers behind, became the emblem of word salad, fodder for western and non-western crony media in endless misogynist caricature – all of which depicted as me in the fraudulent display.
When in fact, I have challenged everyone engaged in the mob ambush against me for an open public debate in world view which has been declined to suit the convenience of evil to subvert and distort my unique identity as it is with all living species in the natural creation.
Not even the free speech birth right to respond to relentless insolence and virulent attacks until now were considered normal with censorship, gag orders and cancel culture becoming pervasive in the authoritarian targeted practice in the name of democracy.
Besides, Who says Hillary Clinton and Kamala Harris are women?
Only then the question of breaking the glass ceiling arise.
I pose the same question that is arrogantly and presumptuously raised to me by power mongering egocentric prejudiced clique anchored on ad hominem misogyny against me having and continue to phenomenally benefit from my existence as a woman.
One receives what one offers another anytime. It is called the Karma with or without any cuss or slur.
You may have the whole world with you in the persistent pursuit to assail TRUTH, nonetheless in vain.
The wicked and crooked devils never prevail over Almighty God’s Will.
Padmini Arhant
Outgoing Presidential Pardons – Beg Your Pardon 🤔
The outgoing Presidential pardon to Anthony Fauci, Liz Cheney and next of kin Hunter Biden within family – distributed more like candies to savor the moment in sober time.
By the way – Isn’t the one guilty of any wrongdoing and crimes seek pardon?
Should the preemptive Presidential pardon to these individuals be then considered they are guilty prompting extraordinary selective Presidential mercy upon them?
Beg your pardon 😱 bemoans democracy!
Padmini Arhant
Gender Orientation – Male, Female in Natural Creation
Absolutely only two genders.
Female (X) and Male (Y) in natural creation.
The exclusive male and female factor respectively is the fundamental fact in procreation leading to generations in life longevity and eternity in Almighty God created “World without an end.”
For that matter, the same is applicable in practical and mechanical designs from plumbing materials to electrical and magnetic settings in the directly opposite format male and female components and as +ve and -ve to charge enabling functionality.
The distortion exerting own complexity and confusion against anyone is a psychological deranged syndrome.
Any other orientation deviating from the natural design is genetic disorder, social experiment and worst of all political indoctrination.
Nonetheless, discrimination of any kind against anyone is unnatural i.e. human developed behavioral misconduct prompted by ignorance and prejudice.
Padmini Arhant
இறைவன் படைத்த உலகம், இயற்கையின் ரீதி – இரண்டு ஜாதி மற்றுமே உருவாக்கப் பட்டது
இறைவன் படைத்த உலகம், இயற்கையின் ரீதி – இரண்டு ஜாதி மற்றுமே உருவாக்கப் பட்டது.
பெண் ஜாதி. இன்னொன்று ஆண் ஜாதி.
சங்கத் தமிழ் இதை உறுதியுடன் தெளிவாக பதித்து உள்ளது.
இதை திராவிட அரசியல் மூலம் கலப்படம் செய்து – மனித படைப்பில் மூன்று பால் உருவாகி – ஆண் பால், பெண் பால் மற்றும் மூன்றாவது பால் தமிழ் சங்க நூல் குறிப்பிடுவதாக அதன் பெயரை குறிப்பிடாமல்,
ஏனென்றால் – அப்படி ஒரு பால் இருந்தால் தானே பெயரை துல்லியமாக சொல்ல முடியும்.
தற்சமய அமெரிக்காவில் தேர்தலில் பலத்த காயத்துடன் தோற்றுப்போன அயோக்கிய கும்பல் சீண்டி தூண்டி விடும் பொய் பிரச்சாரம் பிரச்சார கருவிகளுக்கு தன் முகத்தில் தானே கறியை அப்புவதாகும்.
ஏனென்றால் இதே பிரச்சார போலி குழு திருநங்கையொற்றி அவதூறு செய்தல் அரசியல் சர்வாதிகாரத்தின் மும்மரமாக இருக்கையில், தானே ரெண்டும் கெட்டான் நிலைமை. இதில் தான் இவர்களின் முகமூடியை இவர்களே கிழித்து கொள்வது வெளியாகிறது.
மற்ற படி கடவுள் படைப்பில் – ஜாதி – ஆண் மற்றும் பெண் இவை இரண்டுமே தான் இயற்க்கையின் செயல்.
மற்றவை எல்லாம் செயற்க்கையாகும்.
இவைகளில் முக்கியமாக, மனித உடல் உறுப்பின் கோளாறு, மரபணு கோளாறு, சமூக பரிசோதனை அதைவிட அரசியல் போதனை வதந்தி இந்த செயற்க்கையின் மூல காரணமாகும்.
தற்சமய அரசியல் இந்தியாவின் மத்திய அரசான ஹிந்துத்வ வெறித்தனம் ஒரு பக்கம்,
மற்ற படி தமிழ் நாட்டின் சாபக் கேடான மற்ற தென் மாநிலத்தைச் சேர்ந்த அந்த மாநிலத்தின் மொழி, வம்சாவழியில் வந்த தமிழ் நாட்டை அந்த நாளில் இருந்து இந்நாள் வரை *அந்நியர்கள்* ஆளும் அரசியலால் தமிழர்கள் மேல் திணிக்கப் பட்ட பெரியார் என்ற ஈ.வே.ராவின் கர்நாடகாவின் திராவிடம் என்ற மரபு தமிழையும் *சுத்த தமிழ்* அதாவது பச்சை தமிழ் அடையாளத்தை அழிப்பதற்கான சதி.
மேல் குறிப்பிட்டவர்களும் மற்ற படி ஐரோப்பாவின் அரை குறை கணிப்பு அதோடு சூழ்ச்சியின் காரணத்தில் அந்நியர் கூட்டணியின் இந்த சூட்சமம் எந்த மாநிலத்திற்கு குறிப்பாக கர்நாடாகாவிற்கு இந்த திராவிடம் சொந்தமோ அதை தவிர்த்து விட்டு,
தமிழர்களை திராவிட கால்வாயில் தள்ளி இதுவரை அரசியல் வியாபாரம் நடத்தும் – வாரிசு அரசு, ஜன நாயகம் பெயரில் நடக்கும் கண் கட்டி வித்தையாகும்.
இதில் மக்கள் உழைப்பின் மாநிலச் சொத்தை திராவிட அரசியல் தன் குடும்ப காணொளி நிலயமாக்கி – சூரிய ஒளி, கலைஞர் ஒளி மூலம் – தமிழ், தமிழ் பண்பாடு, கலாச்சாரம், இலக்கியம், சரித்திரம், பூகோளம் எல்லாம் தன் குட்டிச்சுவரான திராவிட மாநிலமாக்கி சின்ன பின்னம் செய்து வருவது, தமிழ் நாட்டிற்க்கு திராவிடம் என்ற சாபம் செய்யும் பெரிய துரோகம்.
இதை தமிழர்கள் உணர வேண்டும்.
பத்மினி அர்ஹந்த்
அழிவின் காரணம்
அழிவின் காரணம்
பெண்ணின் இழிவு அழிவு. பெண் பாவம் பின் பாவம் காலமாகி தற்சமய பாவமாகியுள்ளது.
எத்தகை ஆதிக்கம். பிறரின் உரிமையை பறித்து தன் சுகம் ஏகபோகம் பணம் புகழ் பதவி அனுபவம் அழிவின் அஸ்திவாரம்.
சர்வாதிகாரம் எங்கும் எப்பவும் சர்வநாசம்.
பத்மினி அர்ஹந்த்
Sky News DownUnder👎
The wannabe, opportunist, desperadoes Sky News DownUnder👎 in the rugged outback Australia is literally and realistically DownUnder👎,winning the title unparalleled parasite and groveling sycophant media pit / Outhouse.
Watch where you are going?
Padmini Arhant
“Authentic” Intelligence Paradox 😅
Those behind the trend projecting anyone from anywhere? including others in semblance to the meaning🌷and / or the name ending in ‘ni/ mi’ letters and phonetics importantly shady, nefarious, unscrupulous…elements serving as proxies and pawns of the frustrated desperate despotic megalomaniacs epitomize lunacy, idiocy, fantasy and hypocrisy.
Wanton Provocation – in association with viz. Liz common nick name of E(liz)abeth. Liz is also the prefix in (Liz)ard.
Similarly, Mic(hell)e aptly suitable as Michael.
When such nuances are prevalent – it is indicative of the promoters and practitioners topsy turvy twisted mind.
Not to mention, their names appropriate to them and their profile.
The trait considering them touting their intelligence – unparalleled “authentic” 😂 and promoting the one they are incessantly obsessed with – “artificial” is analogous to the pigs in the swamp running in circle in search of their pig tail. 🤣
The ongoing insanity presided by psychotic sociopaths is the parody and tragedy of this century making themselves and their inventory of cartoons the spectacle of the world.
विनाश काले विपरीत बुद्धि । With the end near the mind reverse and becomes unclear.
Padmini Arhant
Happy Pongal! Happy Makara Sankaranthi! 2025
Provocation, Manipulation and Subversion
Provocation, Manipulation and Subversion using live or the so-called AI model created by destructive factions aimed at “anti-natural endowments and elements” in denial of empirical facts about them as the existential threat endangering lives and human normal existence invoke their termination in the attempt to inflict otherwise on the “obsessed target and their concerned. “
Since world creation till date EVIL never prevailed over God’s will to restore sanity, tranquility and civility in disarray under EVIL and COHORTS ambit. Like all things designed to expire, the EVIL presided tumult is no exception rather on prioritized expiry.
FEI – For Evil Information -Any unnecessary hoodlum tactics and hooliganism however is disposed as garbage.
Padmini Arhant
Originality
No matter what alloys are used to produce value based item and marketed as gold value – they are never the same nor equivalent to the original gold.
Originality has the naturally assigned uniqueness similar to individual DNA in genetic code.
Imitation and Fake never equal original and authentic.
Padmini Arhant
Satans and Proxy Duplicity
Satans and their proxy – pack of hyenas always scavenging for survival being pathetic necrotic parasites.
Stealing zodiac sign and basically any and all positivities ironically from the targeted arch rival never absolve Satans and proxies of abhorrent violent homicidal, genocidal, terror and cannibalism..amongst other despicable pervert lewd crude characteristics laden with con, scam and public summary execution of the rape victim – own member of the race.
As for once again pirating phrases and content from the one incessantly pursued and ridiculed via murderous proxies and sponsors epitomizing cowardice.
When hoodlums, hooligans, perverts and gangsters run the gamut making mockery of civilization and civilized world,
The opinion stated in the sub-domain of this site in Tamil language – that “anyone can achieve the dreams and ambitions in life by honing in the skills and talent they possess together with hard work” is a statement maintained now and forever.
Only the narcissists and shallow minds possessive of any and all natural endowments to all beings at varying degree and potential would demarcate as exclusive to them such as – the bizarre ominous cult Artificial intelligence created by them otherwise touting themselves as authentic intelligence.
When the mob rule could exist anywhere run by terrorists and terror sponsors,
Why is it not possible for sanity, civility and integrity to achieve anything in their life?
Something for the AI dependent sadists to think about if such ability is possessed within.
Last but not the least, the misogynists steer the society against natural and biological heterosexual relation designed by nature for life expansion besides emotional feelings.
Heterosexuality is fundamental in God’s creation of life existence and sustenance as well as in the expression of love and affection.
The misogynists with their given disposition and behavior are instrumental in promoting and practicing relation paradoxical to heterosexual orientation.
These misogynists are also closet culprits while actively engaged against heterosexual norm.
This trait is specific to misogynists with their outlook and overt targeted misogyny culture despite the fact their emergence from the womb and reproducing themselves (God forbid🥹) the requirement of ovum and the uterus.
No wonder such travesty running the show on the world stage fears and detest the termination.
All things are set to expiry – the merchants of destruction and devastation for own economic and political interests in particular expediting the process.
Padmini Arhant
Satan’s Satanic Antics
Satan’s assortment antics eventually backfires at satan. The evil course in diabolical activities suffice in the end justifying the means in the laws of nature.
Satans could subvert anything but not own guilt ridden conscience witness to all of Satans’ satanic activities making the run impossible for Satans to sprint from them.
Shackled in guilt and sinful existence, Satans invariably succumb to own karmic woes sooner than later. Satans obedience to the devil often expedite such outcome.
Satans propose and God disposes viz. Winning Election and warfare makes Satans head spin and detach from torso.
Evil never prevails over Goodness being God’s will.
Welcome to Reality.
Padmini Arhant
Ego is Self-Mortification
One can never reason with humongous ego – emanating from human presumptuousness.
The Supremacy syndrome is the one with plenty of hubris, ample attitude with nothing to guide from within in self-mortification.
The crown doesn’t fit the head circumference given the oversized trait representing the dire strait.
Supremacy and Slavery are intertwined complexity. Supremacy is enslaved to internal negative vices prompting quest for relief via external enslavement in slavery.
Those who can’t help themselves from self-decadence are not the ideal choice for others’ salvation.
Padmini Arhant
FPI – For Public Information
FPI – For public information all inclusive without exception –
My pledge of allegiance ever remains with none other than Almighty God, Natural Environment and republic wherever – humanity at large.
Padmini Arhant
Anchored Deranged Misogyny Syndrome
Mud slinging without basis is own burial in quicksand.
Evidently straightening a twisted dog’s tail is possible than addressing a deliberate malignant identity misappropriation and anchored isolated deranged misogyny syndrome.
Not to mention, the foolish trend led by guilt-ridden white and everlasting victimhood black culture in the vain efforts seeking compensation and compromise for mutual benefits from the scapegoat not even remotely related to neither race nor individual legacy when reality verifies the reverse with the former duo as recipients in the combined patronage.
Similarly applicable to the tail wagging crony contingency from anywhere viz. Indian polity, conventional + social media and entertainment – the undeserving parasitic major beneficiary in newly invented unprecedented demented custom.
Padmini Arhant
Devil is Rattled – Tell Tale Signs
When the devil can’t handle anything in colossal defeat i.e. shellacking in 2024 Presidential race resulting in full blown exposure of the devil’s demonic traits, the devil and the DEI evil conglomerate are rattled simply on fait accompli.
What does the devil and DEI Evil clique do to exhibit original image?
After all, how long could the devil and evil DEI B.S. hide in stolen positive identity and persona of their most envied target and arch nemesis?
At the end of the day, the glove never fits the blood stained hands from emergence in the world to parasitic existence till now.
The devil in particular bolstered by Evil corpses seething in monstrous jealousy, hatred, anger and misogyny…surrender to the demon in their mind and turn berserk witnessed lately in the series of diabolical activities since historic loss in 2024 U.S. Presidential election – no surprise there.
Amongst the devil’s homicidal mania going back to drug peddling childhood rampant in adult “gay hood” preying on own bedfellows black and white especially sodomizing the white male Larry Sinclair on wheel chair to the latest saga involving black female dental hygienist raped deserting a black child from the rape and the raped mother summarily executed using taxpayer provided security personnel by the tran duo were not over.
The latest victim black male personal chef on the paddle boat at taxpayer funded vineyard estate doorstep…merely symptomatic in the otherwise serious personal wreck syndrome.
How could it be possible?
In conveniently deploying the ridiculous race card having been ironically responsible for abuse and murder of own people while promoting Black Lives Matter imposed on others. Meanwhile, brazenly committing heinous crimes from the political immunity sanctuary.
The Presidential election 2024 defeat is avenged with drone drama carried out in the East Coast and of course in California – the anchored domain for the devil and evil DEI dichotomy.
Upon not being satisfied the devil had to orchestrate the mowing of innocent bystanders and crowd gathered at 2025 New Year’s celebration casting yet another devil’s loss of control in demonic outrage in New Orleans, Las Vegas and New York simultaneously at the dawn of New Year 2025.
The devil let loose has been causing bloodshed, terror and carnage at the onset and went overdrive since November 4th, 2016 upon the devil ushered into exit having created tremendous pain, suffering and misery to all those the devil has targeted thus far.
There are two outcomes in the attempts to shape anything for useful purpose.
The iron ore – a rough hard core metal is brought to shape for various usefulness by hammering and / or heating that turns into molten state – the idiom – strike when the iron is hot is precisely for that reason as it conforms to malleability.
The other outcome that fits the devil and transcontinental evil conglomerate is the above outlined process only renders them out of shape and beyond any positive engagement as they are paradoxical to the characteristics and personal nature of the devil and evil cohorts hell bent on – our way or highway even when that is in the bottomless pit.
It is not the question of the devil and evil compatriots on the precipice.
The downfall from the self-destructive provocation for own individual and collective self-serving never to be fulfilled evil aspirations not ever coming into fruition is conspicuously leaving the devil and evil syndicate possessed to own peril.
Somethings never change for better by design and own volition.
The Gospel Truth – the devil and EVIL DEI Dichotomy may presume to hold the gavel amongst own insular world not barring the natural intelligence subversion convoluted as artificial intelligence though hopelessly fleecing on the contributions of the ones despised and vindictively pursued in obsessive disorder.
The Supernatural phenomenon is the ultimate power in the mantel of Supreme Authority as the Cosmic Force since creation of the Universe and galaxy until present into infinity future and beyond.
Padmini Arhant
The Misogyny Irony
The Misogyny Irony
Padmini Arhant
The irony in misogyny is denial of own emergence and existence otherwise impossible in the absence of woman’s reproductive organs.
The spermatozoa without ovum would be extinct is empirical fact. The natural factor.
Rooster in the Hen House – Never mind on what emerged first the chicken or egg even though the emphatic response is egg hatched into chicken. In the course producing roosters and hens by the hen.
What is relevant in the contemporary era?
The hyenas guarding the chickens (roosters and hens) den proven existential threat to own citizens and humanity at large.
As for ‘thirsty’ slang – the same is epitomized and exemplified by the source and catalysts in colonial imperialistic hegemony involving terrorism, oppression, repression and supremacy syndrome.
Whoever says and thinks – narcissists and compulsive impulsive abusers admire their image upon shown the mirror?
Padmini Arhant
Drone King to Spinning Top – Barry Soetoro aka Barack Obama Political Voyage
Drone King to Spinning Top – Barry Soetoro aka Barack Hussein Obama – the Political Voyage
Padmini Arhant
Talk about “spinning fellas” – the one who is unparalleled in this trait and trend – is none other than the one with the real name Barry Soetoro spinning to Barack Hussein Obama.
The one conning citizens from all walks of life asking for campaign donations of $5 – $5,000 and more with fake fraudulent requests including enrollment in Obama University (that’s right fellas and folks – Obama University – Perhaps in the Obama University specializing in con, scam and art of deceit).
The shenanigans in 2008 – 2016 as the so-called community organizer deriding the Wall Street as “Fat Cats” in own lexicon while firmly seated on the same Fat Cats’ lap since debut in politics and multipolar 2007 election campaign and thereafter by selling healthy, hardworking ordinary American citizens to health care industry historic scam in the name of the abominable OBAMACARE.
In return for trading American average citizens in the health care gimmicks with mandatory health insurance rewarding the combined health care industry with windfall absent stipulations on the health industry to meet fair bargains ignored for mutually exclusive financial gains – cashing and capitalizing with mansions, seaside castles, vineyard estates… on treason in quid pro quo in the healthcare scam and several scams throughout 2009 – 2016 and resumed in 2021 – 2024.
These alone besides endless blatant violations of electorate trust and plethora of flip-flops make the spinning top collapse turning upside down witnessed in the U.S.Presidential election 2024.
Something to deliberate and ponder – the process known as Soul Searching for the irreversibly stained pot calling the kettle “black.”
Padmini Arhant
Musk Donald Trump Prez – AI Pick Controversy
The AI pick Sriram Krishnan by the upcoming Musk Trump Presidency.
Hopefully, in the current AI frenzy,
There will be real criminal investigation in the mysterious death of the mere 26-years-old Indian American Suchir Balaji – born and brought up in the Silicon Valley, California.
The young tech AI researcher and whistleblower during employment with the controversial Open AI organization,
Open AI – One of many tech and corporate donors to Trump Inauguration Fund 2025. Open AI has generously donated $1m to Trump Inauguration event.
The young AI specialist Suchir Balaji reported to have died from suicide on Dec 4, 2024 in the common incidents of murder declared suicide having become the norm.
Will the Musk Trump AI pick Sriram Krishnan – if confirmed, would be able to call for thorough investigation in Suchirc Balaji’s murder related to AI controversy barring any adulteration and domestic as well as foreign intrusion to stymie the facts based criminal procedure?
The Lord Sriram Krishnan would definitely demand and investigate to get to the bottom of the slew of Indian origin students and young recruits murdered in the campus and off – corporate sites in the targeted fatalities of American born Indian students and students of Indian origin arriving from India.
The murder incidents of Indian origin students and case like Suchir Balaji is very alarming and disturbing considering the unusual number of murders of Indian origin young talent in this particular year Jan – Dec 2024.
Suchir Balaji’s parents request for investigation has been denied with the matter closed as “suicide” which in itself has blown the lid off the can suggesting more to it than meets the eye.
Indian appointees as CEOs in the corporate world especially in the tech industry and politics is far from merit oriented and tribalism.
In reality, it is deliberate sinister considering the tech and politics entrenchment in secret society and dark age agenda using Indian image and names specifically Ram, Krishnan and Ramaswamy…so on.
There is never a free lunch. Nothing happens without underlying malignancy in the contemporary global ploy.
Suchir Balaji is one of ongoing examples as victims in the year 2024.
My sincere condolences to Suchir Balaji’s parents in the Silicon Valley. May time expedite their healing with peace in the most difficult time of their life.
Padmini Arhant
Subversion of Foreign Religion
The ones who engage in distortion and subversion of foreign religion basic requirements.
1. Correct pronunciation of Hindu Gods and Goddesses names.
2. Stop manipulation and hyperbole convolution of mythology by tossing in your spin ball machine.
3. Know the facts prior to promoting propaganda.
Sri Narayanan is Lord Vishnu who in return is also Goddess Durga in the female form. Goddess Durga never gave boon to demons with immortality knowing they have to be dealt with by her when they impulsively wreak havoc which they always do sooner than later.
Padmini Arhant
திருநங்கை அவதூறு அவரவர் அழிவு சாபாமாகும்
திருநங்கை அவதூறு
அவரவர் அழிவு சாபாமாகும்
பத்மினி அர்ஹந்த்
திருநங்கை அவதூறு அவர் அவர்களின் அழிவு மற்றும் அவதூறை பரப்பும் பிச்சைக்கார பரதேசிகளுக்கு அது பெரிய சாபம்.
புதிய தலைமுறை டிவியோ அல்லது படு குழியில் மாண்டு போயிருக்கும் சாக்கடை கழுசடைக்கும் முதலும் கடைசி கண்டனம்.
கருங்கண்டம் ஆப்பிரிக்காவின் கென்யா அல்லது அமெரிக்காவின் சிகாகோவை சார்ந்த கருமுண்டம் அரக்க அராஜகம், களவாணித்தனம், பிறரின் உரிமையை பறித்தல், விபச்சாரிகளை வைத்து அரசியல் நாட்டாமை செய்யும் அசுரர் விபச்சார அரசியலுக்கு, தமிழ் நாடு மற்றும் இந்திய அரசியிலோ அல்லது அயோக்கிய அரசியலுக்கு அடிமையாக இருக்கும் கூலி பட்டாளங்களுக்கும் இந்த அறிக்கை மற்றும் கடுமையான கண்டனம் சாரும்.
ஊசிப்போன ஊறுகாய், சலிச்ச சாம்பார், துர் நாற்றமான காஞ்ச கருவாடு, செத்து போன ஆடு, மாடு, கோழி, பன்னி – ஏன் உயிருள்ள மனிதரையே தன் வன்மம் கள்ளம் கபடம் நயவஞ்சகம் இவைகளுக்கு இரையாக்கும் இந்த மனிதர் என்ற சாவு கிராக்கிகள் – இந்த ரகம் தான் இவ்வாறு தன்னையே கேவலப் படுத்தி கொள்வது. தேவையின்றி மற்றவரை உதாசினம் செய்வதே இவ் வகைராக்களுக்கு தன் பிழைப்பாகும்.
திருநங்கை என்ற எண்ணம், தூற்றல், பெண் ஜாதியை இழிவு செய்யும் உனக்கும், உன் குடும்பத்தில் இருக்கும் அத்தனை பெண்களுக்கும், சுற்றார், நெருங்கிய மற்றும் தூரத்து உறவு அனைத்து மொத்த உன் குடும்பம், குலம், இனம் யாவருக்கும் அது சாரும் – இது உண்மையாகும்.
அயோக்கியர்கள், அட்டூழியர்கள், அநியாயம் அவர்கைளின் கொள்கை, பேராசை பொறாமை பிடித்து பிறரை ஏளனம் துன்புறுத்தல் இவைகளே வாழ்க்கை வியாபாரமாக வாழும் அனைவருக்கும் –
உன்னுடைய சடலத்திற்கு நீயே சங்கு ஊதி கங்கு வைக்காதே.
உன் துற்செயல் உனக்கு கொள்ளி வைக்கவோ உனது இறுதி சடங்கிற்கு வாரிசு இல்லாமல் சபிக்க படுகிறாய் இந்த பிறவியிலேயே – இது உன் வினை உன்னை தண்டிப்பதாகும்.
பிறரை திருநங்கை என்று தூற்றல் அல்லது உன் துஷ்ட மனதிற்கு எப்படி எல்லாம் தோன்றுகிறதோ அதில் ஈடு படுவது நீ மற்றும் உன் அடுத்த தலைமறையின் அழிவை அலை போல் அலைந்து அனைப்பதாகும்.
உன் கருமாதிக்கு நீயே காரணம் ஆகாதே.
தன் வினை தன்னைச் சுடும் உரட்டியப்பன் வீட்டைச் சுடும். இதை எக்கனமும் மறவேன்.
பொறாமை பேராசை பொல்லாத வியாதி. இதில் அவதி படுபவர்கள் தன்னுல் இருக்கும் விஷத்தில் அல்லாடுவதாகும்.
நீ யாராக இருந்தாலும் வம்பை வீணாக விலைக்கு வாங்காதே. தன்மானம் சம்மானம்.
எப்பொருள் யார் யார் வைப்பினும், அப்பொருள் மெய்பொருள் காண்பது அறிவு.
நன்றியுள்ள பிராணி நாயிடம் நன்றியற்ற பல மனிதர் கற்றுக் கொள்வது அவர்களின் இந்த பிறவியின் முதல் கடமையாகும்.
பத்மினி அர்ஹந்த்
Photo Gallery
Progressiveness – Definition
Maintaining ethical efficacy on all issues which is unfortunately a tall order for many especially those perpetually using and exploiting others for own hideous ideology defined progressiveness or otherwise besides sinister political agenda.
Accepting own failure in personal and political front by not unscrupulously reassigning to scapegoat anyone especially the target not even remotely associated or interested in such activity or position.
Finally, reaching out to blue sky, dark clouds or rainbow with the plagiarized connotation “Wipe your tears” is at best disingenuous and at worst hallmark of self-dissipation i.e expedited exit.
Anything contrived as the proxy exposes the original-self and those behind the charade with nowhere to runaway from oneself. Accordingly, acknowledge defeat gracefully and victory with humility.
These characteristics are categorical regression and not progression by any stretch of imagination.
Padmini Arhant
Identity and Gender Misappropriation Culture
Identity and Gender Misappropriation Culture
Padmini Arhant
In accordance with the prejudiced misogynistic trend targeted ad hominem at me in the past nearly twenty years for;
My tireless selfless sacrifice with unconditional positive contributions – though it originated since my childhood benefitting many across the spectrum in diverse fields especially politics, media, economic and entertainment sector…in unimaginable proportions and magnitude.
In return, only subject and ill-treated with absolute contempt, hatred, envy, caricature, character assassination not barring real life threats on many occasions and pejorative malice in relentless violation of my individual rights and life,
At Almighty God’s will, the following are adapted towards any and all those from wherever in cohort with the prevalent diabolical trend and culture on deliberate identity, gender and personal profile misappropriation of me and my life with utter disregard for my individual rights will now onwards be treated as such.
To begin with;
1. All western claimed achievements across any field and industry since western origin until now and moving forward would be identified and attributed to the western touted latest Africanization trait and deemed African and that of Africa and not that of the Western imprint.
2. In this respect,
United States would be remembered and reminisced as Uganda.
Imperialist Britain already renamed Barbados being the key promoter behind identity misappropriation of me as an entitlement.
Australia – the ever eager lap dog in all western and western White Australia policy would hereafter be known as – Aboriginia – The land of the Aborigine which is rather accurate and hence not identity misappropriation rather appropriation.
Canada – The North American nation as the vassal state of British Commonwealth under the liability known as the Royalty would be identified as – The Central African Republic.
New Zealand – Namibia
Western Europe combined – Equatorial Guinea
Israel – Ethiopia – The origin and the land of Judaism – fitting the appropriate nomenclature not misappropriation.
All those engaged in the hideous provocative misclassification of my gender, identity and personal details will be hereafter individually and collectively recognized as such:
Miss Barry Seotoro alias Miss. / Ms. Barack Obama,
and
Mr. MICHELLE OBAMA alias
Miss. Ms. Mike Obama.
Mr. Hillary Clinton, Miss Joe Biden, Mr. Kamala Harris, Mr. Nancy Pelosi and so on.
Above all,
Miss / Ms. ELON MUSK
Miss. Ms. Joe Rogan
Mr. Tulsi Gabbard,
Miss / Ms. Vivek Ramaswamy – considering the transformation in reality.
Similar nomenclature would be applicable to anyone from wherever regardless indulging in such practice against me.
The proxies, puppets and pawns recruited as me in media. politics, entertainment etc.,by the evil syndicate behind this ludicrous identity misappropriation would be applied gender alteration and transposition practiced against me in the last two decades until now.
In observance and approval of Almighty God’s direct ordinance – Never to tolerate injustice, abuse and unprovoked wanton indignation and insolence that I have been arbitrarily isolated and attacked until now,
The above outlined identity of all those would be relevant with immediate effect.
One receives what one serves others to set the unprecedented convoluted subversion of me, my life and identity straight that is otherwise taken for granted, misused and exploited by the beneficiaries in worst parasitic present toxic environment.
Padmini Arhant
अपनी चित्ता को आग देना
झूटी तारीफ़ और झूटी तालियों के लिए अपनी अभागन जीवन को और बड़ी गाली बनने से बचायें।
बड़े पर्दे पर मुझरा करते करते आदत से मजबूर, उनकी दुर्भाग्य या बतक़िस्मती असली ज़िंदगी में भी उनकेलिए वही पेशा बन जाता है।
जो किसी के कहने पर भी – “सलामे इश्क़” का पैग़ाम पर्दे पर और पर्दे से बाहर मंच पर सज दजकर चलती फिरती साड़ी और जवाहरी दुकान बनकर अनजान की बुराई और दुहाई के लिए निकलती हैं । उमराव जान १ और २ – कोई फ़रक नहीं पड़ता जब लोंभ, ईर्षा और मूर्खता के चुंगल में फ़से इन पीड़ित आत्मावों के लिए – जो अपने ही विनाश और तबाही की घोषणा / ऐलान करते हैं।
हैवान और शैतानों के बहकावे में आकर किसी दूसरे और वो भी जो अपने काम के काम मतलब रखने वाले के सात, यूहीं पंगे लेना अपनी और अपनी परिवार, कुल और वंश की चित्ता को आग देना होता है ।
इसे कभी भी खेल या मज़ाक ना समझें। जिनपर काल मंडरा रहा हो, वही ऐसे काली करतूत पर उतरते हैं ।
उदाहरण / मिसाल के तौर पर – रामायण और महाभारत ही काफ़ी है ।
पद्मिनी अरहंत
Fakery in Politics’ Bakery
Why does fakery stand out in politics’ bakery?
It is obviously stale stock rebuffed with stand out flavors palatable to vendors’ liking rather than consumers’ taste.
Importantly, how does politics utilize own inventory to maintain control despite win or lose election?
By using two or multiple stocks from the same inventory representing the same power mongering controlling clique. They are deployed for the same purpose benefitting the same old establishment and power centrifuge. Nonetheless, assuming diametrically opposite roles purely for optics.
The one female member overtly pushed in the front line as the syndicate choice all the way to the highest position – such as the Presidency though in practice resigned to be the servile obedient puppet to behind the scenes puppeteers never wanting to let go of the control freak enigma.
While the other, deployed as opposition to the political class i.e. the recruiter having recruited the female representative initially as the member of Congress, foreign affairs committee, intelligence committee and variety…and suddenly designated the national adversary asset – such as the Russian asset.
When in reality, the recruit is the political party hired asset to negate the actual target eternally regarded the arch nemesis by individual and collective anti-republic and anti-humanity force.
The pseudo paradoxical recruit is conveniently placed to be the nominee in the most sensitive position like the National Intelligence Director to infiltrate the elected opposition and provide the service in accordance with the contrived situation.
Similarly. the other top positions such as the second-in-line VP breathing over Presidency is a hack to favor those running the gamut in the political game.
At the end of the day, the hyenas can never pretend to be the doe and buck for too long.
It is not in politics’ nature to be true, transparent and trustworthy. Simply not possible by default in politics’ character.
However, the curtains do fall and the show ends to relieve the foul play fatigued captive audience sooner than later.
Padmini Arhant
Ethics – Padmini Arhant Quotes
What is life without ethics?
Shipwreck waddling and wobbling on rough seas and still water ultimately sinking to the bottom.
Short cuts in life are invariably short-lived in any endeavor – fame, fortune and power.
Shame is self-inflicted parody in the effort to shield own reality.
Padmini Arhant
Scams and Thuggish Politics – Origin Chicago ILLINOIS
When thuggish corrupt criminal politics with domain in Chicago, Illinois abuse expired power by permanent tagging, trolling, spying, snooping, eavesdropping as lewd crude perverts misusing taxpayer dollar against the same taxpayer, the perversion is harbinger of self-apocalypse affecting any and all around that are related to the source.
As for those caving in to political intervention in what is supposed to be fair anti-discriminatory practice in society and economic sector such as financial products and services prove their servile complicity with thuggish politics invoke their termination in the kamikaze collusion.
The malicious intent to harm and hurt anyone is precipitous decline surrendered to the devil.
The devil never prevail over God’s will.
Padmini Arhant
Human and Demon Character Distinction
Human character accepts victory gracefully and defeat with humility.
Demon on the other hand indulge in self-destruction with overwhelming hubris upon anything regarded success claiming credit all to oneself and vindictiveness immersed in vengeance in the aftermath of rejection and failure.
Those who dig grave for others invariably find themselves in it sooner than later proven in the karmic woes ever haunting them becoming the corpse while living.
Those who are sulking and seething in rage over the latest 2024 United States Presidential election outcome are challenged to spill their animosity in public rather than deploying slaves as stooges in the despicable demeanor.
As always win we are invincible, lose then you are inexcusable is the demonic trait.
You are that no matter what and whom you pretend to be is exemplified in viciousness expediting your own termination.
Padmini Arhant
The Reality on Impermanence and Charity
Nothing is permanent and never to be assumed invincible without failures. From Napoleon to Alexander and those later Hitler…until the ones since the dawn of the twenty first century till date met their fate.
The one who is the permanent giver such as the Almighty is Almighty for that reason.
Meanwhile the takers as receivers of the benevolence from the venerable join the ranks of those as the recipients of such magnanimous charity.
Piracy and plagiarism of identity and biography is testament to disgust to own profile and epitome of indignation.
Padmini Arhant
பெண்ணின் இழிவு செய்பவரின் அழிவாகும்
பெண்ணின் இழிவு செய்பவரின் அழிவாகும்.
பிறரின் உரிமையை பறித்தல் தன் நிலமைக்கு எதிர் மறையாகும்.
நல்லவருக்கு தீங்கு, நம்பிக்கை துரோகம் செய்பவர்களின் கதி – அவரவர் வினை, ஆணவம், அகம்பாவம், நயவஞ்சகம் ஆகியவை அவர்களின் கர்மத்தின் படி அவர்களையும், மற்றும் அவர் குடும்பம், குலம், இனம் நாசம் நிச்சயம்.
இதை யாரும் எவரும் தவிர்க்கவோ அல்லது தப்பிக்கவோ இயலாது.
இதை எக்கணமும் எப்பொழுதும் மறவேன்.
வினை விதைப்பவர் வினை அறுப்பர். இது இயற்க்கையும் காலத்தின் நீதி.
பத்மினி அர்ஹந்த்
Decadent Politics Expedite Self-Destruction
When politics maintaining the decadent culture to typecast their target as anyone despite the trend condemned as reprehensible are warned about the reality and legality in this obtuse political hysteria.
The target portrayed as anyone however, by default inherit the economic wealth and financial fortune of any and all, the target is suggested and exhibited as them.
It cannot be only the negative connotations the target has to contend with in this moronic vitriolic identity swap / transposition.
The politics and their proxies in their inventory should be mindful of their wayward conduct prompted by uber hubris and chauvinism in this regard and otherwise. Not to mention the wannabe opportunism impetus expedited expiry as witnessed time and time again and contemporarily in all affairs.
The ones flashing power, fame and fortune yet endlessly starved for more with no end in sight as a curse are affected by incurable terminal disease.
Nothing is worse than discontentment serving as the fundamental cause of inner conflict within and turbulent mind.
The poignant fact is the ones promoting and practicing vilification and denigration of woman and womanhood not only exhibit the treatment of women in their family and society in that order.
They also confirm their diminutive stature in their delusional political might that is judiciously diminished and extinguished by Almighty, the creator, protector and destroyer of anything counterproductive especially the ever destructive ego.
Importantly and relevantly,
All are born naked and leave the world as such without exception with a distinction among them many in politics and economic sector live and die Soulless.
Soulless is the worst form of nudity that deprives the subject of humaneness, character and above all self-dignity.
When one respects oneself and prioritize self-esteem in personal and collective behavior, that characteristic is naturally exemplified towards others.
The woman in particular without whom the spermatozoa would be extinct without her ovary.
The emergence in the world for male dominance not possible without her womb.The Hindu epics Ramayana and Mahabharata depicted the extinction of culture and clan in entirety indulging in indignation and insolence of woman and womanhood by overactive male testosterone.
One has to be careful and wise about what they serve others for that would be duly returned to them by their KARMA and whom they incessantly and mindlessly target in their obsessive pre-occupation.
Never too late to refrain from stupidity when choosing enemies in life.
Padmini Arhant
Efficiency Government by Plutocrats?
President-elect Trump has tapped tech entrepreneurs Elon Musk and Vivek Ramaswamy to lead an advisory group focused on cutting federal spending and reducing the size of the government.
Hmmm 🤔 –
Are these gentle ladies duo going to cut federal government spending?
The politics from top to bottom funded by hard working ordinary Americans drowned in generational debts dominated and ruled by plutocracy.
The efficiency government aimed at cutting government spending and waste expenditure would hire two women to head the department while preparing to downsize government?
Shouldn’t one be the example of the Change one wants all around?
Politics and hypocrisy are permanent regardless.
Padmini Arhant
Imposters on Notice
The criminal syndicate of any representation, composition and dimension staged theatrics featuring imposters in the futile delusional endeavor to stymie Almighty God divine mission not barring subversion of my identity invite and expedite individual and collective irreversible apocalypse.
The evil diktat orchestrated impostors as flashers in particular would be instrumental in the imminent and inevitable collapse.
Evil taking anything for granted especially the one they regard arch nemesis is the basis for own and the deployed elimination and disappearance into oblivion.
The Wicked and Crooked especially the power mongering and profit oriented politics anyhow and somehow with proxies, puppets and pawns in disguise will never prevail against God’s Will.
Padmini Arhant
Male Supremacy?
Esquire: What it means to be a man in contemporary American culture?
Response: Recognition and Reconciliation of these things.
Spermatozoa would be extinct without ovary in the likelihood of Asexual viz. Amoeba.
‘X’ is explicitly female chromosome in the genetic code.
Nothing and none are perfect beginning with self.
Pride precedes downfall.
Machoism and Chauvinism are vanity driven to insanity.
All things are set to expiry without exception sooner than later.
Those who dig grave for others invariably find themselves in it.
Above all NONE ESCAPE KARMA.
Padmini Arhant
Frankly Speaking on Poison Politics
U.S.Presidential Election 2024 Outcome Speech Synopsis
Israel – Middle East Conflict – Segment 2 – Extemporaneous Presentation from and by Padmini Arhant
Israel – Middle East Conflict – Extemporaneous Presentation by Padmini Arhant
Identity Politics – Divisive and Polarizing
Identity politics is nothing more than a severe complex among those behind the promotion.
Identity politics eternally tagged to race, gender, sexual orientation and other denominations are best laid to rest.
The connotations maintained and referenced for paradoxical implications in the beginning of the second quarter of the twenty first century make such politics decadent, divisive and polarizing.
Above all, none regardless have the right to continuously deem and define anyone against their expressed will, dissent and objection as anything to suit their interventional pejorative.
Treating others the way one wants to be treated is the basic etiquette that unfortunately, identity politics violates by impulsive disorder.
My message post election will be presented shortly.
Padmini Arhant
A-list Syndrome?
Those touting themselves or undeservingly elevating those from own type as A-list exhibit self-adulation and megalomania.
Not realizing it is your KARMA that determines your listing.
The reality is the presumptuous self-promoting category invariably fall in B-list i.e. BLACK LIST for all the negativities, evil deeds, crimes and flamboyant arrogance under KARMIC recognition ordained by Almighty God.
B-list aka Black List is where such activities and personalities belong and cataloged much to own delusion as otherwise such as A-list.
The malicious intent and indulgence to make the target the laughing stock blow back on those in accordance with;
One reaps what one sows in life without exception.
Eventually, the JOKE is on the ones stretching beyond in malignancy and contempt expediting own elimination in the process.
Padmini Arhant
Identity Politics is Personal Complex Disorder (PCD)
Identity Politics is Personal Complex Disorder – Rooted in Prejudicial Nomenclature
Padmini Arhant
The Supremacists of any genre, Globalists and the so-called Commoners dependent Royalists in Britain and Europe besides politics variety obsession and manifestation in identity politics viz. the race – the black race in particular, gender again trans gender amongst all, sexual orientation fixated on gay and any further declassification of human race is the mirror reflection of own identity i.e. personal complex disorder.
The Obsessive Compulsive Disorder (OCD) in identity politics for these deeply disturbed minds evolved into PCD – Personal Complex Disorder.
The one who does not know oneself often indulge in attributing self-identity crisis to those they envy for their clarity in every respect. Nonetheless, the futile effort to disown individual contra personality syndrome is a wasted life.
The process delivering outcome in revealing them as to
Who they are?
Anything except for being human devoid of humane values and virtues to accept human race as humans barring explicit identification badge depicting isolation and denomination to suit their troubled tarred mind in the inescapable escapism of their identity dilemma.
It is time to bid FAREWELL to such narcissism under the guise of elitism ravenous in devouring anything and everything opposed to their paradoxical nature.
Unfortunately clarification is required on basic to general matter. Let alone anything above average amongst the dull nitwit ignoramus pompous arrogant ignorant recalcitrant decadent sect and political cult behind identity politics mania.
Padmini Arhant
Politics OCD
Politics’ OCD
Only applicable to those indulging in self-caricature.
Politics’ OCD in plagiarism and piracy of positive traits and intellectual content of their arch nemesis viz. me for their personal and political gains further expose such politics’ corrupt, criminal and controversial insignia together with the unscrupulous media – the local and foreign surrogacy in the monkey act.
The crow is not a peacock. Likewise the pig is not and never a GOAT. However, the monkey in the monkey see monkey do – Monkey act is relatable to that of an Ass aka Donkey.
Choose your enemies wisely.
Indian politics and crony contingency – specifically applicable to you as well with a warning to refrain from exploitative, wannabe and opportunistic politics – the worst ever.
Padmini Arhant
Clear the Clutter in your Mind
Applicable to all those delusional personalities and wannabes dwelling in Fools’ Paradise.
Clear the clutter in your mind. You will realize your inherent flaws drowned in fake flamboyance about your identity. Only you can do it yourself.
No maid, toilet cleaner, waiter, flight attendant, fat, thin or any other physical and social economic class…you assign them in your prejudice shackled mind can do it for you.
Despite your world crumbling and relevantly stink without their service . You are proven worthless even otherwise. They are the ones who make you seem important as you flaunt yourself to taunt them.
Ignorance is abyss when marred with impotent arrogance.
Padmini Arhant
FASCISM WRIT LARGE AS THE SO-CALLED DEMOCRAT
FYI : California Franchise Tax Board
Sacramento, CA
For the umpteenth time – Please refer to the third letter dated June 13, 2024 received by you with signed delivery confirmation. The letter with proof of tax payment on August 23, 2023 with 2022 tax return e-filing, bank confirmation on the payment and email references were all enclosed for your reference and settlement on this matter.
Yet, for political reasons the weaponization of taxpayer funded State and federal departments and agencies trending since 2009 – 2016 against then Republican Tea Party by Barack Obama in personal vendetta against them for questioning Barry Soetoro’s birth certificate was responded with terminating the Republican Tea Party in the first term in 2012. It was just the tip of the iceberg.
The trend then continued against political opponent Donald Trump from impeachment proceedings to libel suits with no end in sight.
Similar tactics deployed against me and my family by the same members i. e. The OBAMAS having abused and continue to use and abuse our presence, profile and all things politically and personally exclusively beneficial to them from DAY ONE until now by tagging, trolling, pirating anything positive and impressive for image enhancement despite being warned to cease and desist.
Notwithstanding thousands of $$$ fraudulently amassed from us by the fleecing scammers, abusers and parasitic exploiters till date make them indebted to me personally and my family.
Politics’ underhanded bullying and harassment is sheer desperation in the face of own extinction.
If you can’t express gratitude as the chief beneficiaries of our timeless sacrifice and benevolence, the least you could do is abandon the self-destructive attitude hurting you in the attempt to harm others.
By the way – nothing to do with Angel Reese and anyone with Brown last name in your nuanced attacks directed at me. They are all yours and you can keep them.
That goes for Kamala Harris – the name Kamala adopted from my explanation of my first name conveniently plagiarized for political and fortune harvesting in your end game once again for en masse deception like in 2009 – 2016.
The same resumed in 2021 – 2024 with blatant intention to extend the sham indefinitely with ludicrous WOKISM.
Originality and FAKE distinction is authenticity never in requirement to be anything other than true self unlike the impostor ever remaining fraudsters in vain.
Padmini Arhant
Republic Status in Politics
Politics must realize – Power is granted by the Republic and remain with the Republic.
In contrast, the inherited political culture in Washington intensified since 2009 – 2016 resumed in 2021 – 2024 and attempting beyond, the accumulation of power and wealth among politics leaving the republic with generational debts and endless crises.
Such politics exit is imminent with expedited expiry.
Padmini Arhant
SOS and Wannabe Distinction
The distinction between SOS and Wannabe is self-explanatory.
SOS – Save Our Soul is a cry when someone’s life is in danger typically a request to be saved or they might die in such situation.
There is also likelihood of SOS played out like,
“The Boy who cried Wolf.” – Simply to test the level of attention from anyone or all. Again the indulgence often never without teaching the attention seekers a lesson or two that might even prove serious like for the boy falling prey to own prank.
Wannabe – As the term suggests – they want to be the center of attraction. The publicity starved syndrome by acting out or verbatim usually premised on controversial inflammatory provocative mostly meaningless comments suffice for wannabes to draw attention from anywhere.
It depends on those paying attention to such gimmicks.
Padmini Arhant
Paradox Parody
Crow is not Peacock. Pig is not GOAT.
White and Black playing race card and misogyny ad nauseam is the expedited end of decadence culture with Supremacy guilt on slavery. The latter compensated with affirmative action besides hijacking the positivity and extremely beneficial attributes of the one fleeced on by both without any shame or self-esteem.
The Supremacists’ OCD targeting the formidable female arch nemesis scapegoat the same in the biggest ever blunder to be realized before the defaulters and the crony contingency demise.
The ones who are condemned with excruciating peril and reckoning upon them now and forever hardly have any opportunity other than succumb to self-destruction.
Padmini Arhant
Western Foreign Policy Responsible for Global Nuclearization
Western foreign policy against non-western nations in the 20th and 21sf century until recently is directly responsible for nuclear arms race expanding global nuclearization including North Korea.
Notwithstanding draconian economic sanctions lasting over decades besides military provocation, illegal invasions and occupations in the respective regions.
United States in particular, the pro-Israeli politics on the left and right regardless of democrat and republican in the executive and legislative branch grant Israel indefinite parasitic existence with financial, military, technology and unfettered access to exploit American ordinary taxpayers for Israel’s hegemonic goals and United States strategic dominance in the Middle East.
Not to mention the status quo and tradition since inception and creation of the State of Israel in Palestine is blatant TREASON against United States and the republic.
Israel in continuation of colonial imperialist era is confronted with similar fate of imperialism succumbing to own self-destructive pariah state. The trend in accordance with fire engulfing the origin in the indiscriminate carnage.
Setting fire to the habitat whilst dwelling in it is pyromania gone wild in the self-inflicted end.
Padmini Arhant
Global Energy Corridor – Strait of Hormuz
The Strait of Hormuz – the western side on the Persian Gulf under Iran’s ambit could potentially paralyze the oil exports from the middle east and global production further exacerbating already overrun energy crisis from the middle east conflict and Russia – Ukraine war under the current Biden – Harris administration.
The soaring inflation in the United States would have severe impact from the energy crisis generated in the Middle East meltdown.
All the more reasons to ceasefire on all sides with economic factor being crucial if civilian lives don’t matter to the warring factions.
Padmini Arhant
United States and Israel Expediting Global Nuclearization
Global Nuclearization:
The war in the Middle East has long escalated into deadly confrontation.
Iran and Israel along with United States in the full throttled military exchange is heading towards a precarious situation. The alarming transformation is counterproductive.
The latest developments from Gaza to Lebanon now right at the center of Iran and Yemen with Israel and U.S. administration spearheading the world towards global nuclearization rather than nuclear disarmament.
This is a very dangerous transition that has already prompted Russia – the largest and deadliest nuclear stockpile holder to steer towards advanced nuclear proliferation.
Iran with regional conflict focused on the Islamic Republic could even be driven towards nuclear armament upon the ongoing military aggression not brought to a permanent end.
There are no winners in the brutal aggression against one another except massive destruction of lives and catastrophe unless the madness is reined in with immediate effect.
Eye for an eye would only make the warring factions go blind mutually incapacitating one another in the revenge game.
The power of any leadership is not in fire power turning the target nations and own turf into graveyard. It epitomize tutelage to cowardice surrendered to irrational blindsided cavalier engagement.
Such leaderships confirm colossal failure in resolving crises peacefully. Instead submitted to bloodshed and incessant violence.
History is testament to those who live by the sword die by the sword.
Padmini Arhant
Opportunists are Vultures
Opportunists only surface to grab opportunity wherever and whenever possible. They are swooping vultures that stoop to the lowest for the prey without any effort or labor.
They also feed on the carcasses for survival similar to opportunists’ existence in becoming the face of EVIL regurgitating lies and propaganda as recruits for pittance selling the SOUL.
The truth exploiters on their part are required to divulge TRUTH about the shady backers and promoters behind them and their sudden emergence from nowhere in the Presidential race 2024.
Above all, the mega opportunists are they who never stick their neck for anyone – unknown or known especially when the innocent members from own ethnicity and denomination are mercilessly murdered in college campuses like the several incidents against Indian students 18 – 22-years-of-age were targeted in hate crime from January – April 2024.
The opportunists not surprisingly despite legal background and other influential professions in American society never muddy their feet by lending a voice even to those among own origin falling prey to vicious hate crimes and political malice.
These opportunists at the same time without any shame and conscience are in the forefront to claim various positions and political status asserting the same racial and ethnic orientation.
Typical opportunism ad nauseam.
The devils’ minions and media propagandists hired by EVIL ever remain the slave to appease the wicked and crooked as the only means of their existence sharing the same destiny and destination as EVIL.
Padmini Arhant
Booby Trap and Gutter Mind
The targeted obsessed misogyny and racism epitomize decadent culture.
Cease objectifying woman and womanhood.
Misogynists’ lens viewing womanhood as nothing more than display for voyeuristic sleazy pleasure deploying proxies from own inventory to denigrate female arch nemesis is desperation exacerbating decadence ad nauseam.
Without ovaries the spermatozoa would be extinct.
Above all, emerge from the booby trap clearing the clutter in gutter mind.
Padmini Arhant
Selective Sensitivity to Mental Capability
It is not surprising to see the tidal waves from politics to media surrogates, clannish entertainment and variety coming to own kind aid and indefensible defense.
The Republican Presidential nominee Donald Trump’s name calling of the incumbent VP Kamala Harris as ‘Mentally Disabled’ undoubtedly created a firestorm in the exclusivity club.
The name caller DJ Trump is best to explain.
Nonetheless, the Republican Presidential nominee DJ Trump perhaps reciprocated to similar or worst monickers against self in assortments varying from Hitler to birthday suit depiction of DJ Trump in public square.
CNN exceeding expectations as usual went beyond in dedicating air time to VP Harris defense upon being referred to as ‘Mentally Disabled.’
Interestingly, CNN or MSNBC and diverse media ever remaining the vigilantes to protect the favored political members neither seem to have time nor any flare to the current VP Kamala Harris public slander of Gen Z – 18 – 25-years-of-age calling them STUPID with inability to perform in any and all matter.
This was politics gone wry to say anything against anyone asserted as political might.
In fact, for all practical reasons Gen Z barring exceptions applicable to all samples on mental and physical assessment as well as behavior, are otherwise quite smart and intelligent based on modern Gen Z – astuteness in technology, business and individuality unlike politics’ arbitrary under evaluation of anyone except them.
The VP Kamala Harris statement against Gen Z was not only inappropriate. It also directly implicated the Gen Z parents, teachers and mentors’ involvement in Gen Z lives in some level if not to major extent as such.
Politics’ gaffes and glitches verbatim eventually return to the point of origin like the boomerang.
Never hurts to observe own actions prior to finding reactions objectionable.
Padmini Arhant
Vice Presidential Debate 2024
Governor Tim Walz had the burden to unburden what is and has been of the incumbent administration.
It is no ordinary task given the magnitude of problems related to the current administration with VP Kamala Harris being responsible as the next in line to Presidency. The argument raised as the reason to nominate VP Harris in the 2024 Presidential race as the automatic and preferred albeit not elected choice.
The political party can’t have it both ways.
Contrarily, the Republican VP nominee JD Vance had to simply remind audience and politics on the Trump Presidency economic record and security. As the Republican VP nominee concurred on the fact Governor Tim Walz had much tougher position than his own in defending personal and that of the administration record.
Nonetheless, both nominees performance elicited that members from across the political aisle could agree to disagree on certain or all of the issues though there were convergence on few issues in this debate. The civility and mutual appreciation between the two members were a refreshing change .
The democrat establishment should abandon questioning Trump campaign on election 2020 and demanding response on whether the 2024 electoral outcome would be acknowledged even before the end of election process.
It is the democrat political class who never accepted 2016 election outcome setting the precedence in this regard. Not to mention the democrat establishment discarding own democrat primary in 2024 subsequent to coup d’état against own incumbent.
The democrat administration and democrat Congress members in the House and Senate together with some RINOes perpetually subject the 2016 election winner Trump to politically motivated litany of charges including at least three unprecedented impeachment proceedings wasting taxpayer money for personal and political agenda.On 2024 election outcome question, it is necessary for democrat hierarchy to clarify to the American electorate if they have any preemptive plan in store to be unleashed after election?
The democrat political class and surrogate media repeat demand from Trump Presidency and VP nominee to submit to election result in November 2024 long before the election conclusion only arouse suspicion about their own disposition. When the status quo apply to them given their track record rejecting the American electorate 2016 election mandate favoring Trump Presidency.
Padmini Arhant
P.S. I have only one name and that is Padmini Arhant. Unlike multiple assortments imposed on me to suit any political will, crony media as well as whoever narratives.
Glass House Reality
The despotism prompting incessant pervert lewd crude indulgence hurling stones at the fixated obsessed target as entitlement sooner than later find own glass house shattered crumbling the dwellers beyond recognition revealing the glass house reality.
What one offers others is what one receives in return.
Padmini Arhant
Character Not Color That Ever Matters

Lord Ram emphasized on deeds aka Karma not creed that matter in living and after life.Dr. King – related to that by echoing the content of the character not color that matters.
It’s time to move towards clear aka colorless society and nurture the importance of deeds with merit and virtues instead of remaining stuck in color coding society. Such trend only reveal opportunism with polarization rather than unity and harmony amongst human race.
Padmini Arhant
Jealousy is Poison
Jealousy is poison that consumes the one from where it emanates more than the one it is directed at in life.
Make your mark in life with your feet and feat not using others or at others’ dispensation.
Natural skills and verifiable deeds attract genuine admiration and appreciation.
Real talent is resistant and resilient to emotions arising from inadequacy and insecurity whether within or outside.
The best response to usual and unusual hostility is unbecoming of such personality and let own positive track record serve as testament.
Padmini Arhant
Falsehood and Plagiarism
What happens when the press in collusion with evil syndicate plagiarize data from personal profile and real life despite objection from the source and attribute to recreate the impressive outlook that is otherwise of the unelected choice against democratic norm?
The desperate indulgence release the unpleasant odor to that of a dead rodent in the trap.
The web of lies only result in entanglement raising more questions on the contrived characterization of the unelected nominee.
The leading press description of their backed nominee’s parents’ academic background do not correlate with the unelected candidacy version during the earlier (2019) and latest Presidential debate in 2024.
In 2019 – Presidential democrat primary debate – the present democrat Presidential candidate then recited the yellow school bus rides as not something comfortable citing discrimination.
The daughter of the supposedly academics in elite institution in California then were unable to provide basic means of transportation viz. a car owned by the family to ride to school for their daughter?
Instead the self-touted middle class member had to deprive some other child from lower income family in local community with an economic opportunity (opportunity economy) taking away the space in the yellow school bus?
The enactment neither bode well nor makes economic sense from the daughter of the purported economic advisor.
Again, the unelected democrat Presidential nominee self-described living status quoting mother’s hard work barely enabled to buy apparent first home when the candidate was a teenager, with no mention of the economic advisor father’s financial contribution towards the purchase of new home or upbringing of daughters pose skepticism on all of the accounts due to conflicting statements and flip flop throughout the Presidential campaign since 2019 – 2024.
Naturally, the question which narrative is not false?
Padmini Arhant
Democrat Campaign Slogan – “We are not Trump” is True
The democrat campaign slogan –
“We are not Trump” is true.
Here is why.
Trump Presidency – 2017 – 2020.
Economic Prosperity.
Border Security. The borders were closed barring illegal entry. The construction of border wall equivalent to fencing the home boundary to prevent intrusion and unlawful access.
No Ukraine or Gaza war.
No abrupt unprepared withdrawal from Afghanistan leading to the United States defense personnel (the honorable Gold Star members) falling victims to the Biden – Harris administration’s egregious extemporaneous decision.
Furthermore, the abandonment of taxpayer funded U.S. defense inventory worth millions$$$ left in possession of the Taliban hardline government exacerbating Afghan population conditions especially the women and children at the mercy of radical Taliban social policy.
No political destabilization in South Asia.
The toppling of democratically elected government of the former Prime Minister of Pakistan Imran Khan in Pakistan in February 2022 via U.S. Biden – Harris administration directed No Confidence Vote to unseat the former PM Imran Khan.
Sri Lanka political and economic turmoil literally driven into banana republic status liken to the military aggression in July 2009 leading to the Sri Lankan Tamils’ commemoration of the month as the Black July.
Lately, Bangladesh in 2024 – the former Prime Minister Sheikh Hasina headed economically stable nation was brought into political unrest similar to Ukraine and several other democratic governments throughout 2009 – 2016.
The interference in Bangladesh resulting in the displacement of economically stable government with Washington democrat key members preferred nominee Muhammad Yunus in the form of interim government.
Middle East in crises in semblance with the so-called Arab Spring in 2010 – 2016.
Africa in chaos – repeating the episode from 2009 – 2016 in Libya the contentious Benghazi Gate, Mali and bifurcation of Sudan among several other states turbulence in the region at that time resumed in 2021 – 2024.
No targeted censorship, cancel culture, gag order on Free Speech during 2017 – 2020 unlike the kind experienced in 2021 – 2024 in COVID Vaccine mandate to the latest open statements from the likes of the former Presidential candidate Hillary Clinton to imprison Trump supporters or for that matter the Obamas instructed blockade on election messages coined as disinformation directing the California governor Gavin Newsom to sign the law to that effect banning free speech 120 days prior to and 60 days after the 2024 Presidential election to install the unelected democrat Harris administration under Obamas and Hillary Clinton control.
Above all, the bizarre wokism in the name of woke culture, the fanatical subversion of the normal phrase “creating awareness” on general human suffering and misery from economic sanctions, political unrest, corruption and warfare completely distorted and twisted entirely for political propaganda to suit anti-common sense agenda.
In all of the above, the democrat campaign acknowledgment that they are not “Trump” is accurate making the voter’s choice clear on distinction between 2017 – 2020 Trump Presidency and 2021 – 2024 Harris administration on Barack Obama and Hillary Clinton oversight.
Padmini Arhant
Common Sense
Intelligence makes no sense without common sense.
Common sense is the corner stone for intelligence.
Common sense is real, actual and factual.
Here is how – A bright sunny day🌞is daylight not midnight with the exception of longest day in summer in the Arctic zone making it the midnight sun viz. Norway known as the land of the midnight sun.
Padmini Arhant
Reality Check on Identity
Anyone proud of one’s heritage has the right to do so. As long as it is not infringing on other’s identity feeling the same about their respective parental lineage and ancestry etc.
Importantly, none have the authority to impose their will demanding one should transform and surrender to their misappropriation of other’s identity they regard as their prerogative.
The flawed fraudulent trend I have been subject to over the last sixteen years by opportunists who initially shooed me away having exploited my kindness and altruism when they pleaded for my help only turns out to own exclusive benefit and unfathomable status in life.
The revelation on exploiter’s duplicity is prevalent in denial of own identity by running away from self to escape from guilt and embarrassment.
When time and individual karma based on personal deeds and performance unravelled reality on –
Who is Who?
The ones who rejected the real as unwelcome suddenly changed course and insist on me being them and vice versa which has been debunked as delusional.
Notwithstanding, the critical scientific empirical fact. The one premised on all creations identified by specific DNA that are unique even within own family and serve as testament to individuality regardless of whether good or bad in the relevant life time.
It is similar in physical form. The distinction is conspicuous in the size of fingers on each palm and toes on the feet varying from one another.
The perseverance to bring someone down with constant disparagement and obsession is neglect of own life and lack of intellect divested in own inadequacy and insecurity.
The reality check on identity is indomitable in own physical, mental and emotional attributes with acknowledgment or the lack thereof from within.
Getting own house in order prior to seeking faults elsewhere would be a refreshing start for the lost minds ever engaged in minding other’s mind and all things personal despite being reminded that it is persona non grata.
Not to mention the futile interference impetus such category to be the example of perfection and ingenuity sought in their target.
Padmini Arhant
P.S. To fakes and fraudsters imitating on fashion, clothing colors, style not even sparing eye wear to imitate target i.e. me. I challenge every one of them to adapt the real values in life in imitation of their obsessed nemesis i.e. Yours Truly.
They are;
Never con, scam, cheat, deceive, incessantly lie and violate anyone for yours and / or others’ interests even if your life dependent on it.
Be true to yourself that would encourage you to be truthful to others.
Respect other’s space and rights the way you expect from all.
Never claim anything that does not and will never belong to you.
Never entitle yourself to define and declare your arch rival however you deem only exposing your pejorative character besides self-promotion as the laughing stock of the world.
OMG! Oh My God! 😱
Politics – granted y’all are mighty in power, fame and fortune with massive crony contingency in media, entertainers and the whole gamut revolving around the political circle.
The mighty sparing the Almighty in speeches and rhetoric, jabs and jives without any references to God or the equivalent viz. Godmother would be gracious and exemplify politics transformation heeding people’s concern and not deep state, saboteurs and dark forces diktat from behind or in the front.
As such, the Holy entity God reference is antithetical to politics especially contemporary politics surrendered to all things unholy in the control of human life and destiny beginning with mind in the prejudicial determination of
Whose is real and whose artificial?
The politics exploiting people’s mandate exercise liberty to mock FREEDOM of others as delusional status.
The egotistical politics regardless of political affiliation is self-ruinous ultimately resulting in life wasted in internal self-conflict unable to stabilize and balance own existence by reconciling the obvious deficiency inadequacy within with rational thinking and acceptance.
Reminder: None can replace or substitute one another given the uniqueness in all creations with distinctive features and specific DNA identifying individuality.
Good and bad attributes are the package deal in the identity profile that could neither be transposed to another nor traded as the conditional requirement.
Being true to oneself enables truthfulness all around.
Padmini Arhant
Exploitation – Knows Nothing About Gratitude and Truthfulness

True Color
Anyone who reveals true color in abuse, exploitation and duplicity never to be trusted again. Ruthless Alligator trick kindness and ends up necrotic parasite in what goes comes around.
You are who and what you are – with negative and counterproductive profile and legacy since birth until now regardless of beg borrow and steal strategy to change persona, making you run away from you in desperation.
Such personalities are doomed now and forever with karmic effects weighing down heavily denying redemption and salvation in living and upon imminent death.
Padmini Arhant
Race and Victimhood Politics
The race baiting and victimhood politics citing slavery and holocaust for nearly a century now is a paradoxical paradigm.
The irony with both claiming to be underdogs while one controlling money, power especially politics, economy, media, entertainment and free access to anything and everything.
Similarly, the other on Affirmative action granted preferences in all things that are to be hard fought by those belonging to neither as non-white racial demography despite merit and talent only to be declared artificial.
Besides, the factual presentation on status quo typecast – anti-Semitic and racist with misappropriation in vogue.
FYI for the ♾️ times – South India is not South Africa. South India is in South Asia which is in the world’s northern hemisphere.
South Africa is in southern region of the African continent in the southern hemisphere. The difference in geographical locations alone are distinct. Not to mention the other contrasting cultural and ethnic diversity on the nativity perspective.
The free geography lesson is to dispel stubborn ignorance in political mindset insisting on anything to be something regardless of the debunked myth.
Padmini Arhant
Presidency without Democrat Presidential Primary 2024
PRESIDENTIAL COUP 2024
The Presidency without democrat Presidential primary setting unethical precedence in all matters.
Some students never have to pass exam in school or college like the rest of them. They can cut across the line of scores of eligible members and be seated as the CEO in an organization. If the path is not accessible then they can adopt crooked means such as coup d’état to assert title ownership.
The developments in the democrat Presidential nominee Kamala Harris with vested interests’ endorsements and heavy lifting is nuclear option 4.0 for Barack Obama Presidency.
It also cast irrelevance to rules and laws in society as publicly expressed by the extended Obama Presidency on national Constitution as “mere piece of parchment” and lately demonstrated against democracy as meaningless.
The same coopted as cohort by the self-touted Prosecutor Kamala Harris on transnational crimes while being the trend setter in the domestic affair related to the highest office on land to be handed over to the coup mastermind and operatives upon outcome.
God Save United States of America! SOS 😷
Padmini Arhant
End of Parasitic Era
The dark diverse forces are reminded on the end of parasitic era.
When you declare yourselves the authority on intellect and wisdom, Why bother demanding plans and ideas from the so-called artificial intelligence?
Dig deeper and dwell in your deranged syndrome on misogyny while fronting a woman as the nominee even there exploiting the nemesis much to recruit as proxy and recruiter’s ruin and peril, to flaunt limping ego(test)tical megalomania.
Empty vessel make more noise.
Padmini Arhant
Make Men Great Again Quest!
Make Men Great Again Quest!
How?
Acknowledging nothing and none are perfect beginning with self. 😳
Padmini Arhant
ABC News Network
What is ABC News Network lacking and lagging as a result in popular rating?
ABC News Network learning the ABC.. in journalistic ethics and objectivity would do wonders in improving the rating. 🙃
Padmini Arhant
Natural Effect
Propaganda brings down the propagandists to the point of no return after exhausting its course.
Similarly, spewing venom incessantly at the target consumes the source post depletion with nothing to protect itself from imminent hazards within and in the environment.
The Kingdom of lies, deception and dishonesty denying other’s rights and life ultimately crumble from the extensive damage inflicted by own doing.
Plagiarism, identity theft, pseudo performance…is escapism in self-embarrassment, ineptness and insecurity typically running away from self to be another by forgery. The actions, nonetheless instantaneously expose the desperate fraudsters and fakers’ futile engagement.
The irony is such indulgence often presumed convincing to captive audience. However, the witnesses to duplicity i.e. inner conscience besides the all knowing all seeing Supreme Light that dispels darkness remain the constant reminder to the guilty shackled in sins of their sinful existence.
The outcome representing the natural effects.
What is served to others is received in fair and equal proportion if not more in accordance with laws of Karma.
It is wise to be mindful not to ignore the inevitable from own action.
Padmini Arhant
X – Phenomemon
The letter “X” deliberately to denote the trans mania only expose the obvious obtuse syndrome amongst the presumptuous self-proclaimed genius of the geniuses declared the authority on intelligence and intellectualism.
The natural science however has one and only representation for the symbol or letter ‘X’ and that is female chromosome in the genetic factor while the male chromosome denoted by ‘Y’ chromosome.
The all out efforts and involvement in the tran propaganda to isolate and demonize the one and only nemesis for the existing diverse cartel and syndicate further affirm X as feminine with clarity. LOL 🤣
The more those who involve in denial and disapproval of the one they incessantly target, the individual and collective instant elimination of every one of them is imminent.
Padmini Arhant
Provocateurs and Narcissists
Provocateurs
and
Narcissists
Padmini Arhant
The opportunists, wannabes and parasites recruited by provocateurs and narcissists fleecing indefinitely on me, whom they continue to abuse and exploit besides stealing my identity and karma are not surprisingly pi**ed at own reflection in the mirror. These are the types and variety who copy me due to lack of originality in anything positive and productive.
We have nothing in common whatsoever and yet they want to latch on despite being told to cease and desist. Perhaps these lots as bullies don’t understand the meaning of being told permanently to lay their offense to rest. Obsessive compulsive violators much to own detriment.
मान न मान, हम तेरे हैवान ।
Translation: Whether you consent or not, we remain the satan harassing you.
To the villains and Satans from wherever;
What you receive from me is what you relentlessly serve me as your entitlement.
Since, I don’t accept anything that I never asked for nor appreciate wanton provocation and imposition of any kind that I’m forced to endure as my fate especially from those having nothing to live for except digging grave only to find themselves in it sooner than later.
I duly return anything back to such category (even this reference is stolen by them to attack their foes using me – pathetic 😱 ) where it belongs appropriately. 🙃
Accordingly, the response is fair and equal to yours – no more or less.
Padmini Arhant
Stupidity in Plain Sight
😳 The political mob tailgating their arch nemesis, literally on the heels since 2008 until now were to find out the latter residing on the moon,
Would they then schedule the flight of the jettisoned incumbent to land on the moon rather than California?
The gesture apparently bidding farewell rather explicit forced exit in overt coup d’état on the incumbent in Washington D.C. to arrive late night in California after the gala opening of the political charade in Chicago is quite telling on narcissism antagonism upon being declined parasitic existence now and forever.
The beg, borrow and steal category is relentless in self-defeating and self-deprecation endeavor.
The developments further delineate authentic intelligence from the proponents and determinants of the artificial intelligence fad.
The nonsensical egotistic psycho drama is stupidity in plain sight.
Padmini Arhant
Parasitic Existence is Toxic
The end to the era of parasitic existence is expedited with toxicity overwhelming tolerance. Anything exceeding the norm face termination in natural and unnatural settings.
Padmini Arhant
Healthy Living
Live own life not other’s life.
Don’t unburden own negative (-ve) burden on the other in return for the other’s +ve value. The exchange of -1,000, -1,000,000,000,000 (1 trillion) or -♾️ dumped on the other would only result in negative to you.
Make sure you are prepared to receive prior to what you offer the other in deed. Your actions are never without consequences at any and all given time.
The laws of physics on gravity, motion, energy, magnetic field…on any surface viz. land, air and water have delivery mechanism based on the type and interaction.
Padmini Arhant
Plagiarism and Imitation
The disturbed mind prompting wannabe opportunist indulgence using nemesis +ve content and unique style in comeback only make them their rival’s obsessed followers in the lack of originality from within.
The obsession with anything and anyone beyond control is runaway character from own life i.e. escapism rather than resolving own conflicting personality and identity complex.
“You are that who you think I am. I am not that who you think I am and insist as such in your prejudiced judgment.”
Clear the clutter in your mind and Free Your Soul for this is your only chance with memory of this lifetime. The failure to do so burden the Soul deprived of peace in living and beyond.
Padmini Arhant
AI Propaganda – Desperation Beyond Salvation
The return of Daily Show host Jonathan Stuart Leibowitz alias Jon Stewart prior and ongoing surrogacy to elites, the classic narcissists responsible for ruining, maiming, violating, annihilating…millions of lives in the abuse of power in public office and duplicity explain two things about the defended and defenders for monetary exchange – intelligence and character.
Not to mention the die hard cronyism with a price tag numbing the brain cells and blurring the vision fail to see the bare facts and acknowledge events unravelling in the irrefutable status.
The intelligence or the lack thereof evidently the false propaganda on Artificial Intelligence about the nemesis place the propagandists in spotlight.
The artificial superficial democrat Presidential nominee on borrowed materials, stolen positive identity as well as image launched by the corrupt league upending democracy in the Presidential race 2024 is déjà vu.
The 2024 Presidential race is the repeat of 2008 Presidential scam with Nobel Peace Prize handed in advance. The recipient in return exacerbating war mongering, violence at home and abroad, toppling democracy (like experienced now in 2024) then for neo-Nazi and military junta governments worldwide, terror and cannibalism sponsoring…adequately highlight the 2009 – 2016 disastrous legacy.
Not surprisingly, the trend vehemently defended and promoted via Jonathan Stuart Leibowitz aka Jon Stewart and alike in plain sight.
Never mind the apathy towards millions of victims’ plight when cheering and rooting for compulsive violators on display.
In character, Jonathan Stuart Leibowitz aka Jon Stewart embarrassing moment brought to light not long ago in the real estate evaluation similar to determination of others’ intelligence as Artificial while presumptuously declaring own Authentic.
Jon Stewart benefited by 829% ‘overvalue’ of his NYC home sale even as he labels Trump’s civil case ‘not victimless’
By the way,
Why on earth the so-called genius prodigies charismatic the messiah…😂 endlessly remain the wannabe, opportunists, impostors and copy, imitate, troll, tag, stalk, haunt and taunt the after all Artificial intelligence despite the warning to cease and desist?
When you think you are exceptional ?🤣
Why is there hostility and antagonism for non- involvement alongside stealing profile of the one you devalue as worthless with Artificial intelligence to attain the pinnacle of success?
What does that say about the intelligence behind such animation?
Go figure if you can?
Padmini Arhant
Padmini Arhant – Gaza War and Rafah Air Strike

Padmini Arhant – Identity Clarification
South & South East Asia – Geographic Location Subversion
Since when the nation in South East Asia and another in South Asia got relocated to EGYPT and Greece?
Perhaps in the mind of those hell bent on imposing anything about anyone to suit their whims and fancies regardless of objectionable offensive trend considered their entitlement in defining and declaring others life and profile as they deem fit.
Granted geography is not everyone’s cup of coffee, herbal tea, masala chai, latte, mocha or Cappuccino😇
However, claiming the Sun does not rise in the East and sets in the west never make an iota of difference to the Sun. It rather highlight the dimwit and derangement behind such obscene obstinacy of subversion and distortion of reality.
Padmini Arhant
Phonies, Floozies, Fraudsters – Harbinger Own Funeral
Phonies, Floozies and Fraudsters deployment harbinger own funeral and that of the participants in the end justifying the means without exception.
The same apply to all recruits, recruiters and especially propagandists with malicious objectives inviting own peril including the source in Kamikaze engagement.
Those who dig grave for others invariably and incontrovertibly find themselves in it sooner than later.
Don’t sweat over your arch nemesis with your racist sexist envious remarks as the former is not on the knees with a rose in hand pleading for attention as you desperately desire much to the contrary rejecting your hideous idiocy and violent repugnant legacy laughing at your hypocrisy.
Prior to making judgment on anyone about their physical looks, make sure to focus on your mirror reflection, relevantly inner-self within known as introspection. That would be the real tell all about human nature – the true natural beauty that ever matter in living and thereafter.
The evil obsession is only guaranteed self-destruction.
The criminal cabal can no longer protect nor defend evil ad nauseam devouring lives wherever possible prior during and subsequent to emergence in politics.
The web of lies spun on social media, main media network, commercial propaganda and politics from anywhere in evil’s favor only further expose EVIL and diabolical agenda.
One can’t run away from oneself and the sins in particular reminding from within on the imminent self- termination.
Padmini Arhant
Electorate Base Rejection in Presidential Race
When either major political party presumptive Presidential nominee or incumbent running for re-election select the second-in-line rejected by the respective electorate base in the primary resulting in failure of the latter Presidential bid due to electorate non-approval, that combination is doomed from the get go regardless of the front liner position as witnessed and experienced in the Democrat incumbency and VP choice.
The candidacy declined by own base with voters clear message not desirable and importantly declared ineligible evidenced in their failed Presidential contest, yet forcing down the choice on them and the rest of the country is indicative of selection pushed through foreign and domestic vested interests categorically undermining the republic and national interests.
Furthermore, the identity and crony politics are best abandoned and instead replaced with legitimate meritocracy to serve the people and the nation at large with efficiency and ethical efficacy that are conspicuously missing and would exemplify the Presidential nominee respecting the republic will rather than political hatch influenced by incognito domestic and foreign intervention trending thus far.
Similar stunt displayed in Britain with choice of the people dismissed and ignored to appoint feudalists globalist servile appointee.
When politics defy people, that setting inevitably bear severe voter backlash in governance and subsequent local to Congressional election.
At the end of the day, it is the ordinary electorate funding these positions as taxpayer and not elitism in disguise.
Padmini Arhant
Padmini Arhant – PadminiArhant.online

Ramadan Greetings – Gaza War Message in Arabic and English – Padmini Arhant
Ramadan Greetings – Gaza War Message in Arabic and English – Padmini Arhant
Scoundrel Kavodhi Funeral
Pakistan Army & ISI Political Intervention – Padmini Arhant
Pakistan Army & ISI Political Intervention – Padmini Arhant
Pakistan Sovereignty Violation
Pakistan Sovereignty Violation
Pakistan Politics 2024 Addendum – (Urdu Presentation) Padmini Arhant
Pakistan Politics 2024 Addendum – (Urdu Presentation) Padmini Arhant (You Tube)
Pakistan Election 2024 – Presentation in Urdu – Padmini Arhant
Pakistan Election 2024 – Presentation in Urdu – Padmini Arhant (You Tube)
Gaza – Rafah Air Strike Feb 2024
Gaza war Rafah Air Strike Feb 2024 – Padmini Arhant
Gaza War Speech – Padmini Arhant
Independent Palestine – Padmini Arhant
Independent Free Palestine – Jan 2024 Speech

New Year 2024 Greetings!
Peace to Free Palestine
The Dumbest Trend
Attention: The Runaway from Self-Identity
Applicable to only those engaged in this dumbest trend.
When you are all shunning your original identity as to who and what you are obviously due to shame and embarrassment of your actions and indulgence in your life,
What makes you all think, dumping your unwanted undesirable identity on whom you incessantly target much to your individual and collective peril experienced by you all at the moment, that would be acceptable to the one you impose yourself?
The dumbest trend not only exemplify your delusions but also your honesty in rejecting yourselves in recognition of your worst profile and corrupt trajectory that even you can’t apparently live with and doing everything to disinherit as to who you are.
Nonetheless, bear in mind (if you have one?) no matter what you do to yourselves to change anything about yourselves, your identity related to your DOB, age, biological, physiological, psychological and sociological and above all your karmic woes cannot be altered, traded and abandoned in this lifetime.
As for your imminent departure from the world, you can’t even get rid of your sins / bad karma as it is the only certainty that accompany your soul to deal with the judgment that determine your fate then onward.
Don’t sweat over the craziest conduct as it only proves your lack of self-respect and disdain as to who you are?
Accordingly, the apt response to identity imposition is;
Your identity is your problem.
The identity swap is ludicrous and criminal that are taken into account against you during judgment upon your inevitable exit from this world.
Padmini Arhant
Gaza Genocide
Anti-Semitism?
Racism, Sexism, Fascism, Narcissism as ZioNazism could no longer hide behind anti-semitism.
Padmini Arhant
Who is Who?
India – Recognition of Dr.Ambedkar ( The society and politics designated untouchability and outcast – Dalitnama / Dalithood Stalwart).
If you truly acknowledge Dr.Ambedkar then Indian politics collectively would uphold the Indian Constitution to the letter – the Charter authored by Dr. Ambedkar to abolish hierarchy with denominations aka abhorrent casteism with untouchability as the norm in society till date.
There would be no communal divide based on religions and majority minority status that is virulently prevalent now surpassing predecessor Congress in this trait.
Indian politics would denounce fundamentalist ideology not embrace and execute the draconian policy.
As for the one claiming to be the caretaker of Africa would not have sold Africa to the military industrial complex enabling militarization of the continent at ease in the greed for power with dismal prospects on re-election to office in 2012.
The so-called empathizer of black Americans would not have turned the back whilst holding the highest office on land with the power to pardon denying the black American Troy Davis falsely convicted of a crime he did not commit and yet sent to the gallows that could have been averted exercising the black Presidency executive pardon otherwise expended in anything proven destructive during the two terms in office between 2009-2016.
The final straw with the member of black race – Miriam Carey – the woman raped and subject to summary execution expending taxpayer dollars in the crime.
Then the child born out of this crime was tossed into the arms of a complete stranger of white race by the one touting blackness and Africanism in the desperate attempt to damage control. Not to mention the black child deprived of a mother and parental care in the offender resigning to deadbeat fatherhood.
Last but not the least, the human nature is characterized as good or evil not from PR campaign and massive propaganda – the culture normalized in politics and other fields in society.
Instead, the goodness and evil is exemplified and verified in actions, decisions and legacy abusing or utilizing power and economic social status to either harm or benefit individual or collective members in personal lifetime.
Vying for undeserving eternal name and fame by politics and alike in the exploitation of anyone and others misery is megalomania and opportunism optimum.
Padmini Arhant
Evil Prima Facie
Quit wicked crooked politics once and for all. United States and cronies led by India’s wannabes, opportunists, traitors, fraudsters, impostors and desperate sycophants – don’t humiliate yourselves anymore in the identity theft charade in subservience to evil connivance, manipulation and subversion. No matter which side of the mouth you drool from, you are evil’s prima facie in the incontrovertible duplicity.
Padmini Arhant
Despotic Politics Duplicity and Impostors
Attention: Despotic Politics Duplicity and Impostors
Padmini Arhant
Despotic politics desperately involved in image salvation and legacy management must realize the incontrovertible fact.
Who were at whose doorstep knocking and begging for help to achieve their unimaginable political dream in their life?
Then immediately pulled the rug under the feet of the same member and family having utilized their monetary and intellectual contribution with no credit or gratitude whatsoever. Instead volleyed mockery and relentless public indignation.
Notwithstanding the vile vengeful vendetta targeting the benefactors of their political ambition with ruthless tactics and hostility prevalent until now much to own and progeny peril.
Even the Cairo speech was plagiarized and lifted with no mention or attribution to the original source delineating the much propagated artificial intelligence from authentic intellect and importantly human character.
As for wannabes, opportunists and parasites chosen by evil to challenge Almighty God’s ordain and will,
Where were you all having been around in public domain related to politics, beauty pageantry, entertainment, religion and economic sector etc., long before my public emergence?
Even though, I have been approached long before by politics – the proof provided in this website from the predecessor former President George W. Bush, the message from the Democrat President Jimmy Carter and family and several others throughout my lifetime.
Did it not occur to any of the evil nominated designees whether as politician, entertainment and beauty pageantry or religious zealots, media and assortments from India or anywhere regarding their fake fraudulent divinity and divine orientation flaunted at evil’s behest and orchestration?
Politics and entertainment in India, United States and colluders – those representing both and / or either for glory hogging via exploitation and manipulation of my hard work and excruciating journey which they have contributed to in the worst form exacerbating my plight and that of my family are the two sides of the counterfeit coin.
Indian politics and entertainment are required to shun futile publicity stunts to resurrect own long deceased tenure once and for all.
If you really cared about anyone other than your obsessive self-adulation, you would not be tone deaf to human suffering in your domain and anywhere for that matter, which you are responsible for in your complicity with evil.
Importantly, where were you all hiding and in hibernation when Sushant Singh Rajput, the actor and rising star fell victim to Bollywood and Indian political exploitative tool and forced to commit suicide.
None of you could even bring your phony selves together to express remorse at Sushant’s premature demise and many like him subject to disappearance through your active and passive suppression of anyone not in your corrupt criminal league.
Interestingly, now you are vying for cheap publicity at my personal achievement entirely at my intense hard work and many sleepless nights over these years, relevantly without being paid a dime by anyone or monetary compensation ever.
Instead depleted my personal savings and assets until now, to which rather than gratitude contrarily vilified and denigrated by you and your kind having extorted me and my family for your personal aspirations in life.
Let this be clear none of you have any association or relation to my individual feat except you together as active obstructionists and protagonists never missing any opportunity in evil contrived script to make me anonymous much to your collective fait accompli.
Stealing others good KARMA bear unbearable consequences affecting even the progeny in inheritance.
Never claim or seize anything that does not belong to you is the norm in any civilized culture besides the cardinal rule in the cosmic ordinance.
Quit your ravenous craving for undeserving fame, fortune and power at other’s misery.
Stop fooling yourselves and the world at large. The show ha been long over.
In doing so, your karma shackle you all and your descendants in indebtedness for many lives to come to those against whom you commit unforgivable sins in your surrender to evil.
Shame on all of you above mentioned and anyone treading on self-deceit trail only to experience self-inflicted embarrassment and humiliation.
The celebrity related content referenced below strictly apply to only those fitting the description and wanton provocation flaunting long dead celebrity status.
The publicity starved variety dying for attention in the constant harassment of their she-nemesi through crass stunts, exhibit strong admiration for Hitlerism where entertainment industry is prime propaganda hatred machine.
These cohorts in their consolidated cowardly nuanced charade.cheering Hitler in following Hitler indoctrination via entertainment viz. the cinema and any available outlet much to every individual and collective tumult in the league is desperate times losing sanity and propriety.
Celebrity from the big and small screen in the United States, India, Britain etc., especially the one from American TV sitcoms, Britain origin talent show leading panelist, the British chef.. from the west and,
Indian film industry all the woods combined from Bollywood, to Tollywood, Kollywood, Mollywood.. your subservience to appease evil conglomerate in your persistent misogyny, she-bashing and she-indignation,
Meanwhile stealing my original natural positive demeanor in your pseudo mannerism as yours only exemplify your cheap conduct laden with bruised ego and inner lacerations.
Never forget you reap what you sow in life and inherited by your next in line.
Every one of you and your accomplice in any shape and format in the evil enacted theatrics are cursed, condemned and denied redemption for your deliberate willful indulgence premised on greed, envy and humongous hubris.
Above all, every one of you in this collaborative evil actions and connivance earn only gloom and doom beginning now lasting eternity with direct impact on your immediate and next generations due to your mendacious volition.
You may individually and collectively engage in fraudulence as impostors in mass deception of yourselves and the entire world but,
Not the one who is all knowing, seeing and cognizant of who is who, from where and what about them…details to minutia since their Soul existence and the journey now and beyond.
Padmini Arhant
Conflict – Synonyms
Conflict is the synonym for war, discord and warfare.
This is for those, who through IT cell recruits keep referencing “Conflict” in the titles in publications on this website and sub-domain alluding to be incorrect.
काला अक्षर बैंस बराबर ।
Padmini Arhant
Celebration – Bright, Beautiful & Picturesque
Lord Murugan Holy Symbol and Meaning
Israeli Palestinian Conflict – Part 2
Israeli Palestinian Conflict – Part 1
Beautiful California - Yosemite National Park
Lieutenant Young Wu United States Air Force and Padmini Arhant - The Pilots. Flying the plane over blue skies in California, Sept 5th, 2018 was Awesome.
Beautiful California - Genesis Point
Holy Shrines Tour
Gallery
Image Glimpse
Gallery
Goddess Durga
Swastika – The Sacred Symbol – All is Well

Grim Reality
Grim Reality
What is the difference between politics, religion and science?
Politics is fiction in perpetual denial of anything inconvenient and truthful.
Religion is faith suggesting trust and belief in the force primordial.
Science resigns to fact from experimental data yielding positive or negative results not without margin of error either way.
Unfortunately, the three are intertwined with politics misusing religion and science for political gains and adverse cause.
Religion is politicized to assert dominance of one against another.
Science gets political within scientific domain and in dismissal of religion as hypothesis.
From Cradle to Graveyard – Despite lining the coffers at others’ expense, none taken in the coffin leaving behind even skin and bone.
Greed, envy, animosity…array of negative vices consume the hosts in the end justifying the means dilemma.
Padmini Arhant
Organic Intelligence Erring!
The ones flaunting the so-called organic intelligence with constant jibes at the allegedly artificial intelligence could perhaps seek assistance from the latter in spelling adjectives correctly to make sense of the message. Even basic spell check feature would serve the requirement.
The country is indeed precious not prescious than any individual, entity, political party, institution and organization.
Needless to say, it is applicable all around without exception.
Unlike the trend, smothered in hypocrisy beckoning others to do what is apparently unacceptable to selves to the extent of blasphemy.
Padmini Arhant
Small Minds Interpretation of Matter
The interpretation of anything as small, average, little, dwarfism etc. is small minds’ diminutive status – uncomfortable and unable to deal with own reflection and reality.
Let’s examine history on animal and humans of towering height and the fateful outcome.
Tyrannosaurus rex aka T—Rex – 12 feet in height – twice the size of 6’ human, also known as the fierce carnivore devouring prey upon contact finally became extinct. The T-Rex left behind tyranny derived from Tyranno-saurus nomenclature and traits for comparable humans in height and behavior.
Among humans – The Ugandan former President Idi Amin 6’4” spared none in violent atrocities and barbarism against own people. The tall African leader tallness diminished in proportion with monstrosity against own citizens made to flee for life.
The internationally known terror organization Al-Qaida leader Osama Bin Laden branded the terror mastermind was also 6’4” and engaged in activities 12 feet under detonating explosives that eventually led to the bottom hole in life.
The former Iraqi President -Saddam Hussein 6’ dwelling in different palaces ultimately sought refuge literally in a rat hole.
There are more examples in this regard. The world continue to witness and endure such characters’ abnormal sinister indulgence producing the drastic curvature or decline whilst living and legacy.
Suffice to say tallness is not greatness and not all tall people and species possess qualities and characteristics matching or exceeding their physical height.
On the contrary, the above members and alike are the proof as to how tallness stooping to the lowest of the low in deeds and thoughts hurting and harming others can contract their size into nothingness and many forced into oblivion.
The actions guided by the mind and intellect in rationalizing right from wrong, goodness from evil, kindness from violence etc.define the greatness or littleness of human identity.
Additionally, remarks such as little people and other connotations are the result of envy emanating from self-inadequacy, deep insecurity, extreme inferiority complex lacking in self-esteem.
Padmini Arhant
The Gospel Truth
The Gospel Truth – The Cardinal Rule on Sinful Life and Legacy
Padmini Arhant
The color of the sky cannot be changed to suit and fit the preference of the beholder. Synonymously the skin color in a given life remain the same regardless.
Getting over color code – in blackness, whiteness, brownness, yellowness, pinkness, orangeness and any other shades and racial tones…merely an exterior skin layer with color of the blood (RED) remaining the same and uniform in all living species i.e. humans and animals alike, the realization in this context is the preliminary step towards civility.
The life history in terms of social identity – color, creed, education, economic, political background etc., ending and the memory of it erased with the termination of life in any particular lifetime except for the accumulation of karmic debts pending settlement, the condition is irrefutable.
That being the fact of life, what’s all the fuss over melanin and skin pigmentation and other factors?
The Soul’s unknown journey though markedly premised on individual deeds aka Karma in any life duration, the hangover on skin color and stereotype are sentiments inhibiting positive thought process and relevant progress in mind evolution.
The efforts instead turned inward in clearing clutter within and deep inside to develop tolerance, acceptance, acknowledgment extending respect and understanding even with differences and disagreements on issues create better relations in human existence.
The humanness rising above prejudice, lingering bigotry, hatred, subjugation, subordination and discriminatory practice is the greatest challenge for those unable to release selves from the self-imposed constraints on social description.
Learning and striving to be human in nature and behavior is the laudable milestone one could achieve in living and thenceforward.
Having said that, it might be possible to straighten the dog’s tail but not those set and sworn to be unruly and recalcitrant at own peril only to realize the outcome echoing what was ignored and too late for recovery.
On the question of feeling insignificant, it would matter only when such assertions from distinctively insignificant have any significance whatsoever.
Claiming credit on anything that do not apply to self aka wannabe alternatively piracy while shunning responsibility on everything that is squarely own obligation with name written all over is called opportunism.
Financial bankruptcy caused by malicious forces abusing position besides conning and mocking endlessly about the situation long after recovery is sadistic pleasure, the epitome of narcissism
Juxtaposed, personal permanent irreversible moral and ethical bankruptcy drowning in doom and gloom is far worse than any temporary loss in life.
Not to mention the status depleting core human value, irreplaceable element with any monetary or material compensation.
The designee viz. the Vice President as the so-called heart beat away and next in line to the highest office on land selling the Soul to devil for title in name only and price tag is sycophancy traded at the cheapest fire sale otherwise insolvency of brain and human dignity.
The bargain involving self-caricature in ever high ecstasy and delirium on display admittedly from substance abuse evidently never in short supply in the latest cocaine saga at the premise of the most important public office in the nation is sad and tragic affirmation on self-destruction beyond salvation.
The self-inflicted deterioration to appease the devils in disguise is classic example of greed and lack of intelligence resulting in dysfunctional performance and dystopia.
As for the mainstream network with insignia as the sly scavenger FOX represented by hosts and anchors salivating on the bait along with counterparts on curbside communication outlets trading journalistic ethics for demolition of democracy peddling Satans’ satanic nuanced scripts are components of the large fraud machine with engine running on recycled fabrication and distortion in epic proportion.
The combined collaborative efforts expedite their cataclysmic destiny already in progress with precipitous decline together with the entire apparatus behind the gamut on the brink of collapse.
Accordingly the futile nonsensical defamation via VP clown and other pawns sponsored by media cohorts’ directed at the target to convince imaginary audience in empty theater is the ultimate parody glaring at those in fools paradise.
Sins and crimes committed with willful malevolence is demonic despotism.
The characteristics encapsulate brazen conviction to harm, develop attitude entrenched in hatred, contempt, prejudice, misogyny, intolerance, insolence, slur, slander, smear, besmirch, condescendence, patronization, envy, gluttonous greed, hubris, arrogance, bruised ego, violation of rights through invasion of life, privacy, space, perversion, spying, snooping, prying, preying, predator instincts and critically identity theft, identity swap, identity misappropriation manipulated as identity appropriation, isolation to anonymity, exploitation, unlawful intrusion, imposition, harassment, bully, mob mentality, obsessive compulsive disorder with the target…are some of many diverse character deformity and deranged personality laden with ignorance.
Such indulgences bear serious consequences in living gripped with grief and guilt and upon demise leading to the solo journey after life saddled with unsettled karmic debts excruciating thereafter.
Furthermore, lies, deception, dereliction of duty, debauchery, corruption, criminality, treason, violation of trust, abuse of power, public office, people’s money, authorizing censor speech, cancel culture, proxy, pawn, puppet schemes, divisive polarizing poison politics triggered by power mongering and atrocities of any kind exacerbate the inevitable outcome.
Importantly, misinformation, disinformation, falsehood, fakery, vicious propaganda, fraudulence and indignation to name a few…via collusion, complicity, complacency and participation in any capacity with the wicked and evil face enormous Karmic ramifications denying the Soul redemption and peace.
The culmination of all of the above is self-destructive comparable to an object engulfed in sea of fire reduced to ash in the realm of nothingness poignantly reflect the degenerative act dissolution.
The crucifixion of Jesus Christ presumptuously regarded victory proved to be in vain. Those who authorized and participated were pummeled and paid the price nevertheless much later.
Likewise in other faiths, the demons wreaking havoc were appropriately dealt with reminder never to provoke nor challenge the wrath of phenomenon ordained by the creator of the universe.
In Almighty God’s Supreme Kingdom, Justice might be delayed never denied.
Karma effective from the moment wrongful sinister deeds onset, the Soul is shackled in perennial suffering with no respite for the foolish enslaved to evil traits and ceaseless sins.
The cardinal rule firmly apply without exception to all those in violations enunciated above granting none any exemption nor concession.
The Gospel Truth since the dawn of creation established by the Supernatural power remain irrevocable and immortal in essence.
Padmini Arhant
Identity Clarification
Who is Who? – Devils’ Doomsday
Current Events June 2023
United States – CBDC Fiat Currency
Parasite – Parasitic Existence
Parasites unable to prolong parasitic existence are obviously pis*ed. Too bad.
Get a life and quit parasitic culture.
Applicable to parasites who know no other way than colonizing space, home and even personal family conversations.
The brute parasites are desperate like raccoons raiding anywhere. The offenders’ pathetic life prompting them to be perverts – sneaking, eavesdropping, trolling and tagging to own peril.
Shame on you. You are done. So deal with it.
As far as I’m concerned, parasites are persona non grata. Adios! Get lost for ever.
Padmini Arhant
Puppet Politics Clarification
Puppet politics clarifying their exact role to their electorate disclosing the pact between them and puppeteers with price tag for being on the side show alluding to be me losing to the chosen candidate in the Republican Primary and,
Similarly in the national election would help the electorate from wasting time listening to other candidates merely placed for sadistic pleasure to mock me.
The ones behind the charade are reminded that in their game, it’s only them and their team are involved in the self and mass deception.
The portrayal of anyone among them as me losing to their chosen candidate in the Primary and final race is lofty misogyny and prejudice making a monkey out of their not so smart strategy.
With such ludicrous enactments in play to directly humiliate me despite my absence in the race or politics, the response to these shenanigans has the following narrative playing victimhood.
“The mean tweets like the bull in the China shop.”
No surprise here.
The oppressors’ following classic stance is immutable.
Never mind what we do to you. How we behave towards you and treat you and your family like described above.
We will never accept or tolerate your right to self-defense as long as we rein control over everything including your life and destiny.
And I say to that – Wishful thinking.
Padmini Arhant
O’Bummer ! – Vulgar Polity
O’Bummer ! Vulgar Polity trending in the so-called Fashion & Entertainment charade exhibiting self-mortification and caricature of one’s own culture and society.
No surprise.
Padmini Arhant
Lessons from Life – Tamil. Video
Personal Attacks Upon Presentation of Facts
Politics and surrogates’ personal attack via corrupt media and outlets upon presentation of facts is admission of guilt in the offensive defense of indefensible action.
Padmini Arhant
Media Propaganda Purpose
Propaganda purpose is to lie deceive and mislead gullible viewers and captive audience.
Those burdened with skeletons in their life, resign to propaganda for public distraction.
FOX HOAX Media and alike lead the propaganda mission heading towards precipice for them and those whom they promote and defend in vain.
Padmini Arhant
Politics Epitome
Politics epitome of abuse of power, crime, corruption, terror, harassment, propaganda, murder, public lynching, political witch hunt, violence, hate mongering, identity politics, mass deception, lies, exploitation, extortions…otherwise utter lawlessness and fascism authority in power overt ruling or incognito via puppet is existential threat endangering nation and citizens’ survival as a free society with liberty, dignity and opportunity for all barring exceptions in prejudice and preference of any and all kind across the spectrum.
Padmini Arhant
Politics Face
The hunger for power making politics dirty, ugly and nasty while maintaining corrupt, criminal, crass class intact.
It’s a matter of time the power battle within will end politics for good. The incognito and sycophants together in particular.
Padmini Arhant
Get over the 🤚🏽✋✌🏼✌️✌🏿 🙏🏽🙏🏻🙏🏽🙏🙏🏿 mania!
Get over the mania!
🙏🏽🙏🏻🙏🏽🙏🙏🏿

 mania as that is not going to deliver what is aimed at and desired in vain except confirming your asinine indulgence.
mania as that is not going to deliver what is aimed at and desired in vain except confirming your asinine indulgence.Besides in politics such as Indian corrupt criminal signature trait is exclusively yours and no monkey see monkey do act can change it for it is attached to you like skin and bone.
Padmini Arhant
Gossip Mongers’ Life
Gossip Mongers’ Life
Padmini Arhant
How do those who gossip waste their life?
GOSSIP
G – Gas bagging
O – Objectifying
S – Sow Hatred and Spew Venom
S – Seek sadistic pleasure
I – Indulge in indoctrination
P – Poison self and society with vicious propaganda
Padmini Arhant
Gossip – Tell Tale Signs
Gossip –
Tell Tale Signs
Padmini Arhant
Gossip – whisper and weave tales to suit own corrupt mindset.
Who indulge in gossip?
The ones who have no life of their own and become obsessed with another in the nuanced stupidity and insanity. Notwithstanding the target having no relation, connection and / or interests with gossipers and their concoction of characters whatsoever.
What prompts gossip?
The idle mind being the devil’s workshop preoccupy in rumor, propaganda and sleaziness leading to self-humiliation.
How to deal with gossip?
Like with any negative human trait, ignoring gossip mongers to submerge in their dirty swamp would indicate their wasteful life. Furthermore, gossip mongers feed on reaction to ignite more sparks without realizing the flame ultimately engulf their evil act in the fire they started.
Why is gossip the preferred choice for sticky beaks?
They have no life. They seek undeserving attention. They are sadists deriving pleasure minding other’s life to escape from own miserable boredom with nothing to live for and their existence a proven burden without meaningful positive purpose. They are self-declared and exposed excrement contaminating environment.
Those who gossip are controlling and the devils are always agitated upon being rejected for what and who they are. Not surprisingly they accuse the target as controlling for declining to bow to their egotistical narcissism, incessant misogyny, prejudice and hostility.
Gossip and gossipers meet their fate accordingly with a life depleted of self-respect and human value.
Padmini Arhant
Identity Theft
Identity Theft – reiterating earlier statement that I’m Padmini Arhant. My name is Padmini Arhant and not whoever wherever whatever name they choose for me that would be my identity. Such charade only confirm politics in fools paradise.
I say to Indian and international polity – the incognito in particular presiding over the ignoramus identity swap imposed on me as their prerogative.
I need no more namkarans – christening with any names of your choice or random naming which is.a serious violation of my individual right in identity theft mania ultimately hurting all those involved without exception in severe karmic effects.
Padmini Arhant
Toxicity
Toxicity
Toxins in environment are lethal for polluters and general society.
Toxic people waste their life spilling toxicity around. Little they realize the toxic emission from them severely impact them more than anyone else as they precipitously decline in thoughts, speech and deeds. While they make extreme demands on others via speculations about their target’s life and lifestyle without having a clue about any individual’s SOUL, they completely ignore their life letting their mind wander aimlessly firing shots simply to prove their pseudo dominance.
In toxicity, the impurities and contaminants condensate resulting in highly flammable and explosive substance that is certainly neither good nor safe for the toxic producers.
Toxic people are escapists avoiding inner conflicts within and rely on negativities as comfort zone.
The best way to deal with toxicity and the sources is to live one’s life to the fullest enrichment and endearment with those who share unconditional true love and respect.
Living life on own terms with no regrets or apology is an effective distillation process on toxicity.
Padmini Arhant
CNN – Cult News Network
Politics – Fait Accompli
Indian incumbent Prime Minister, Home Minister and so-called political opposition in India kow-towing to international clique, their masters in global arena are reminded once and for all on status quo between you and me and that is explicitly fait accompli.
Accordingly politics squandering public taxpayers money in pseudo elections against me to serve megalomaniacal egotistical political aspirations is lunacy reflecting desperate times seeking desperate downfall – the current stark reality.
Padmini Arhant
Bollywood – Pirate, Plagiarist and Parasite
Bollywood the cheap pirate, plagiarist and parasite – the hoodlum serving hooliganism in politics and international criminal cartel in many aspects, you are long dismissed as irrelevant together with your masters instigating self-embarrassing comments and conduct.
Corrupt criminal politics using you for nefarious unscrupulous purpose further explain your disposable status.
Padmini Arhant
Envy
Envy
Envy revealed in incessant personal assaults, insults, insinuations, insolence, venom, vitriolic condescendence, misogyny, she-bashing and abuse among several self-destructive traits from the source and catalysts aimed at the one and only target as the arch nemesis and sworn enemy responds accordingly.
Needless to say such exhibits are mirror image of them battling with own identity crisis.
Nonetheless, I, Padmini Arhant appreciate the gratitude extended in above enunciated manner by those having benefitted from my tireless contributions and endless sacrifice with God as witness to my sincerity and integrity.
I regard the envy from all those envious members regardless of their titles, designations and so-called powerful influential status – the greatest compliment to me.
The so-called VIPs and VVIPs obsession with me expressed in their relentless personal attacks against me is a proof of my importance and relevance to them despite their flaunted social status.
Envy is regurgitation of low self-esteem, deep insecurity and failure to accept oneself and others with respect. The outbursts and constant disparagement through envy is reflection of inner turmoil.
Those who claim to have everything in fame, fortune and power having nothing honorable is self-inflicted tragedy and incontrovertible reality.
Envy leads to agitation and self-indignation of the origin and agents surrendered to negativity and irredeemable sins.
You and your kind hate me without contentment and yet you all deceitfully engage in identity appropriation of me – want and claim to be me apparently alter ego. There is no doubt about ego in abundance amongst claimants and definitely not my alternative.
The trend is the biggest irony and testament to questionable mental status of yours and those serving ill-fated cause.
Padmini Arhant
Padmini Arhant with Divinity Shiva

New Year Greetings 2023! Padmini Arhant
Bombed Bollywood and Indian Cinema
Message for flopped bollywood and Indian cinema.
Your time has long been up. You are done with having proved your worthlessness in talent and intelligence.
On the contrary your adeptness in narcotics, sleezyness, prostitution, criminality not barring homicide, terrorism, inciting communal violence causing deaths and destruction, arms dealing, sex trade, pedophilia, extortions, exploitation, seething envy, gluttonous greed leading to black money hoarding in offshore tax havens and above all fleecing on others as pathetic parasites in identity theft, piracy and plagiarism galore is nauseating.
The so-called beauty queens of the 90’s and thereafter sleeping their way around unable to make a career prompting parasitic existence is a tell all about celebrity status stripped of shame and dignity.
Indian cinema and politics marred in corruption, scams and scandals are primary sources for accumulating and liquidating dirty filthy money bankrupting local and national economy. Importantly these two together along with other sectors responsible for superficial soaring inflation all year round triggered by unaccounted illegal money in circulation in the economy.
Bollywood and Indian Cinema – you are dead and buried for now and in the future. Get lost.
Accordingly no use flogging the cadaver for the carcass would only serve corpse scavengers.
Never mistake everyone for Sushant Singh Rajput.
Padmini Arhant
PS. This message is also applicable to all parasites wasting life in parasitic existence. There is no respect nor integrity when you and the kind fleece and ride on others’ back as free loaders and worst of all the satans’ slaves taking marching orders from them for undeserving hall of fame, fortune and power which never stick regardless due to fake imagery and fraudulence.
GET A LIFE OF YOUR OWN AND LEARN TO LIVE WITH DIGNITY.
Padmini Arhant
पते की बात
पते की बात
चोरी और ऊपर से सीना ज़ोरि बुरा है तो उससे बड़ी अपराध उस चोरी को उचित प्रचार करना अघोर बेईमानी है। ऐसा पाप जन्मों का भोज बने आत्मा पर दण्ड बनता है।उस देश और देश वासियों के लिए गंभीर पीड़ा और कलंक बने युगों तक पाप में लिप्त होता है।
इसमें गर्व की कोई बात नहीं हालांकि व्यक्तिगत और कुल व्यवहार के तौर पर इस प्रकार की हरकतें शर्मिन्दिगी और अपमानित जनक है।
आख़िर में,
बुरी नज़र वालों और कुकर्म दोषियों तुम सब के मुंह काला।
बुरी नज़र वाले तेरा मुंह काला |
पद्मिनी अरहंत
Celebration of Lord Krishna

Badra Kali – Goddess Durga Incarnate
Badra Kali – The incarnate of Goddess Durga – God Shiva’s divine consort.
Badra Kali – the revered and feared incarnation of Goddess Durga / Parvati
Badra Kali is the force that force evil evacuation and elimination from earth making the world a safe and secure place for all beings.
Badra means secure and safety
Kali -remove. the one who engage in the removal of evil.
Padmini Arhant
Celebration of the birth of Lord Krishna – the incarnate of Lord Vishnu
Jubilation and celebration on the birth of Lord Krishna as Janma Ashtami, Gokula Ashtami aka Shri Krishna Jayanthi is a great sense of joy and bliss.
Personally, being born on Janma Ashtami/ Shri Krishna Jayanthi – the birth of Lord Krishna, on the auspicious occasion – I have the pleasure and honor to extend my gracious greetings and celebratory moment to followers of Lord Krishna – the incarnate of Lord Vishnu.
Happy Shri Krishna Jayanthi / Janma Ashtami / Gokula Ashtami !
Padmini Arhant
Sri Lanka Crisis – Tamil Eelam Desam 2022 – Presentation in Tamil
Duplicity Exposure
Daylight Dakosla (HOAX) – दिन दहाड़े ढकोसला
Claiming a cardboard cut out moon as having plucked the moon from the orbit is Daylight Dakosla (HOAX).
कार्डबोर्ड से कटे हुए चंद्रमा को आकाश की कक्षा से चाँद को तोड़ने
का दावा करना दिन दहाड़े ढकोसला(HOAX) है ।வானத்தின் சுற்றுப்பாதையில் இருந்து சந்திரனைப் பறித்ததாக அட்டைப் பெட்டியால் வெட்டப்பட்ட சந்திரனைக் கூறுவது பகல் புரளி.
Padmini Arhant
Fake Fraudulence False Persona
Fake Fraudulence False Persona
The current trend peaked at false projection amongst those claiming to be the one that is appealing and interesting with desperate efforts in mimicking and adapting appearance, style, hairdo, language and manners…anything and everything is weird classic duplicity. Their indulgence confirm they are mega opportunists and wannabes who are running away from them and their unflattering legacy ranging from corruption scandals, criminality to controversies as persona.
If they have problem in acknowledging them as who they are, what makes them assume their deception would be an exception?
Obviously, their real identity is too embarrassing for them to accept prompting faking and fraudulence.
Little they realize leopard changing spots to stripes do not become a tiger and neither a hyena feigning a fawn become as such.
Being true to self and others is indeed a tall order for them.
Padmini Arhant
I’m glad that India is not my birth place.
It is least surprising on Indian politics and so-called entertainment at rock bottom considering the assassination of a freedom movement leader Mahatma Gandhi following independence from colonial rule which contemporarily reinstated with proxy regimes loyal to colonial system.
Lord Gautam Buddha born in Lumbini in Southern Nepal. Lord Buddha’s Buddhism celebrated in the Himalayan kingdoms and South East Asia.
I’m glad that India is not my birth place.
Padmini Arhant
Corruption and Mob Rule
Acquiescence and appeasement towards politics and corrupt forces facilitate mob rule and tyranny.
The lawmakers as lawbreakers with corruption scandals and criminal records representing law is the irony of all ironies in the so-called democracy with the strings held by unscrupulous elements from behind reining control.
The dominance and Supremacy as feudalists impose vassal statehood on sovereignty and authoritarian rule denying free republic governed democracy.
When democracy and individual freedom is suppressed, the devil claims to be Deus and wreaks havoc. Ultimately the terror unleashed abusing power at the helm succumb to own totalitarian turmoil in the end justifying the means with Karma taking effect.
Padmini Arhant
Author & Presenter
PadminiArhant.com
Prakrithi.PadminiArhant.com
India – Supreme Court Ruling on Godhra Pogrom in 2002
The Godhra pogrom against innocent civilians largely muslims, Hindu and other denominations in 2002 undergoes political spin by the central government headed by PM Narendra Modi and HM Amit Shah. In this regard, the victim widow Zakia Jafri’s plea against SIT ruling on the violence involving Narendra Modi is propagated as the case dismissed by the Indian Supreme Court.
However, the so-called dismissal verdict by the three-judge bench headed by Justice AM Khanwilkar, Justices Dinesh Maheshwari and CT Ravikumar do not bear the signatures of neither of the three justices in this apparent ruling.
The unsigned court ruling – the omission of three SC justices signatures in this ruling confirm the verdict not delivered by the Supreme Court.
The SC’s three judge bench remark quoting “the Indian democracy is under threat” verbatim and subsequently the court ruling delivered without signatures from any and all three of them confirm the decision on vindication of the accused is null and void.
Accordingly, the misconstrued vindication of the accused viz. the former Chief Minister Narendra Modi, Home Minister Amit Shah and others listed in Mrs. Zakia Jafri’s court filing is invalid.
The accused in this matter remain relevant for further legal investigation and proceedings now and in the future.
“What is the importance of a signature in any document?The purpose of a signature is to authenticate a writing, or provide notice of its source, and to bind the individual signing the writing by the provisions contained in the document.”Padmini ArhantAuthor & PresenterPadminiArhant.comPrakrithi.PadminiArhant. comभारत – राजनीति और समस्या
भारत – राजनीति और समस्या
पद्मिनी अरहंत
देश के सर्कार और उत्तरदायक यानी प्रधान मंत्री को पांच गंभीर प्रश्न जो उत्तरदायित्व है।
१. देश पूँजी पति के हवाले करके, उन्ही के सेवक बने व्यक्ति प्रधान मंत्री?
२. देश की सुरक्षा में घटौती लाकर, देश को देश के बाहर आक्रमि चीन के हवाले करने वाले देश के चौकीदार ?
३. और तो और आक्रमि देश चीन को आर्थिक भंडार भेंटकर आयात (इम्पोर्ट्स – imports) के ज़रिये, चीन की आर्थिक स्थिति मज़बूत बनाकर फ़िर उसे भारत पर आक्रमण करने की सुविधा देने वाले देश के अर्थशास्त्री और शुभचिंतक नेता और सर्कार?
४. इस प्रकार कानून बनानेवाले प्रथम कानून तोड़नेवाले बने और वह एलान करें की वही रहेंगे ५० वर्ष से भी अधिक देश के मुख्या यानी प्रधान मंत्री और वो भी लोक तंत्र गण तंत्र प्रजा को ताना शाही में दबाकर अपनी राजनीति कूटनीति जमाने की योजनाएं में जुड़े तत्पश्चात?
५. आखिर देश लोक तंत्र है या केला गणतंत्र (बनाना रिपब्लिक – Banana Republic) जहाँ संविधान नेतृत्व में जन राज्य बदलकर एक अकेले व्यक्ति के इशारे पर उनके क्षति (damaging) निर्णय के चक्रव्यूह में फ़स गया है ?
धन्यवाद
पद्मिनी अरहंत
लेखिका और प्रस्तुत करताPadminiArhant.com
Prakrithi.PadminiArhant.com
Message to Humanity – Padmini Arhant
Spread peace and engage in non-violence. Speak truth with courage. Heed your conscience. Respect life. Serve your nation and people with honesty and integrity. Espouse human values and exemplify in deeds with care and compassion. Voice concern over injustice. Be part of the solution and not the problem. Love mankind and environment for universal harmony.
Peace is Eternal Bliss.Padmini Arhant
தமிழ் புத்தாண்டு 2022 நல் வாழ்த்துக்கள்! Tamil New Year 2022 Greetings!

Words of Wisdom from Venerable Gautam Buddha
Asian Culture - Greeting
India – The Truth Behind Agnipath
Identity Misappropriation
The evil among myriad evil doings are persistently engaged in identity misappropriation. In their evil mind they will define who is anyone and who they are and should be to suit their evil opinion. Contrary to actual evidence.
Anything repeated as a cliche to enforce identity misappropriation as appropriate confirm meaningless asinine indulgence. The identity reality nonetheless proved otherwise is the irreversible revelation much to evils’ indigestible predicament.
Evil bear consequences to evil charade and constant concoctions to validate evil dominance. Besides desperate fabrication, falsehood and fraudulence glaring at combined evil conglomerate bringing mega embarrassment in fait accompli is the fact.
Evil struggle on identity misappropriation is like insisting on 2 + 2 = 5 , 3, 7, etc. but not the empirical 4. The evil obtuse farcical nuance is nothing more than attempting to trap air in the open space or writing on running water. Both clarifying the losing battle for evil and contingency.
Padmini Arhant
Monkey See Monkey Do!
Charity Begins at Home
Who is Jashodaben Narendrabhai Modi?
She is the estranged wife of Narendra Modi, the prime minister of India. The couple were married in 1968 when she was about 17 and Modi was 18. A short time into the marriage, her husband abandoned her.
The RSS led BJP government in New Delhi revoked the Triple Talaq (Divorce) against Muslim women primarily for political mileage to gain Muslim women votes in the state and national election.
The tradition was criticized by Hindutva Prime Minister Narendra Modi as primitive while having deserted own spouse Jashodaben Modi with no alimony or any lawful compensation.
Apparently, the Hindutva ideology practiced by RSS and Indian Prime Minister neither believe nor subscribe to ‘Charity begins at home.’
Padmini Arhant
Imitation
Imitation is flattery. Imitation of positive and profitable values without due credit to the source is forgery – criminal and condemnable act.
Padmini Arhant
The Fly on the wall
What should one do with the fly on the wall?
Swat or pat was the question.
Obviously the former. The flies spreading preventable disease sometimes even causing death is a biohazard. It is important to understand the breeding ground for the flies to survive and thrive. Accordingly the flies are dirty pests and putrefy upon contact.
Padmini Arhant
Human Trait
Human Trait
One might straighten dog’s tail but not certain human trait – the ones hell bent on remaining crooked and wicked in particular.
Spying, snooping, eavesdropping, surveillance, perversion via invasion of home, personal space and life to detect anything illegal. Never mind the illegality of such indulgence. The wrong doers in search of wrong doings waste own life on earth.
The specialists in personal attacks whether direct or innuendo using proxies from own inventory leave an indelible mark about their personality. They are as follows – weakness, insecurity, dishonesty and guilt…among myriad complexities in them in handling truth about them and their actions in life. The relentless personal assaults and individual profile distortion against their target is predominantly related to own inability to defend indefensible track record.
The worst human behavior is starting and causing the crisis. Subsequently grandstanding the crisis without an iota of integrity. Again amidst obnoxiously slight human intelligence with no regrets or remorse.
Exploitation – Who is to be blamed for exploitation?
The exploiters or the exploited?
Both. As long as the exploited allow exploitation to continue with no end in sight, the exploiter would exploit normalizing the trend. The exploited realization of the abuse ending exploitative situation is a direct and effective response to exploitation.
More on the way on human trait.
Padmini Arhant
Skin Pigmentation
Skin Pigmentation
Soooooooo (infinity) hung up on skin pigmentation!
The lighter version under the sun goes tan.
The darker tone in snow and frigid temperature without much sun exposure gets pale.
What really matters in life?
The genuine personality with natural identity – the one not borrowed as imitation or imposed really matter and authentic.
Similarly those who have tasked upon them to define and distort anyone whom they target in culture and identity appropriation – recreating anything against reality only reveal desperation and futile discrimination.
Human character with basic values – treating self and others without pride and prejudice suffice shedding any complex either way – superior or inferior.
After all underneath the light, tan and any shade skin layer, the color of the blood is the same – Thank God.
Otherwise there would be more dirt and filth digging in human indignation.
Be yourself without any burden to impress or appease anyone.
Padmini Arhant
Light and Sound Effects
Light and Sound Effects
The brilliant sunlight – the source of energy and life. Importantly dispels darkness.
The braying donkeys produce noise pollution.
The light and sound effects in this regard are clear in their relevance.
Padmini Arhant
Restoring Freedom?
Restoring Freedom?
Those who embark on restoring freedom beginning with their domain would add some credibility.
The definition of freedom is liberty to choose in life. Having a choice to accept or reject anything unfavorable is the real alternative.
For instance, the solar panel lessee not having a choice with payment options other than the one and only method set up by the electric car company acquired solar power installation. Similarly, binding the lessee on long term contractual agreement with no exit plans.
Last but not the least, not addressing emergency situation during heavy rain and stormy weather resulting in roof leaks from poorly installed solar panels causing property damage.
The necessary precautionary measures from home owner like tarp on the roof even protecting the leased solar panels to avert substantial property damage including solar panels and subsequent liability to solar panel lessor i.e. the company involved in the matter are not reimbursed and expense declined as not the company responsibility.
These issues clarify the status as anything but freedom.
In light of such reality, it would be fair to say the customer in such unfair and unreasonable deals are the ones affording the company’s much touted philanthropy in a war zone besides contributing to extravagant hobby such as space travel.
These activities made possible by none other than customer tied to company policy in direct violation of contract to resolve issues related to leased items such as solar panels.
The corporate overture on restoring freedom would be appropriate upon being made available to customers in practicality in own ventures and enterprise.
Needless to say getting own house in order prior to engagement elsewhere is the dignified priority.
Anything otherwise is open to interpretation as publicity to enhance individual fortune.
Padmini Arhant
Truth about lies and liars
Truth about lies and liars
Liars live in denial. They conscientiously decline reality due to lack of integrity. They dwell in falsehood. They rely on fabrication and fraudulence. They try to run away from themselves despite being prisoners of guilt with unwillingness to introspect and accept fact within.
Soul searching is persona non grata – unwelcome for them.
Instead they lie to themselves and the outside world. In doing so, they voluntarily deceive themselves and others. They are also users, abusers and exploiters as a result. They scapegoat anyone they think are a fair game to defend their indefensible offense, deception and criminality.
They are obsessed with external facade rather than internal cleansing alleviating burden on soul. Those who lie to get anywhere in their life are usually miserable cheaters and knowingly live a dishonorable life.
The Truth about lies is lying traps the mind, pains the heart and ultimately chains the Soul. The choice to be a free spirit is lost in living and dying for them.
Padmini Arhant
Hope, Hoax and Hell
The commonality in Hope,Hoax and Hell besides numeric order is hope proved a hoax turned into hell in events best laid to rest and never to return to create any more havoc.
The analogy is – expired products relaunched in the same package to revive HOPE is a hoax adding insult to intelligence.
Fool me once shame on you. Fool me twice shame on me.
Padmini Arhant
Inconvenient fact on cause and effect
The human nature commonly evade responsibility.
The worst trend is to conveniently ignore the cause and selectively react to symptoms and end-result. The case in point is the global pandemic.
There is absolutely no interest in addressing the controversial game of function research, the investors collusion, WHO and Beijing’s massive coverup on the origin and transmission of the lab manufactured deadly pathogen and subsequent mega profiteering from vaccine and related ethical and scientific violations…
Instead the entire focus is on the effects such as anti-vaxers and politicized pandemic reports.
Similarly, the incident in a gala entertainment event – the choreographed and highly dramatized delivery via scripted monologue – the sly innuendo and insinuation having become the trademark of tacky exhibit is deliberately ignored with exclusive attention on the superficial reaction.
Never mind the origin i.e. the cause. Only rebuke the effect in show and tell action following the condescension.
It’s a matter of what fits and suits the corrupt and prejudiced mind.
Padmini Arhant
Beauty – Meaning and Definition
The real beauty lies in the heart, mind and soul purification reflected in deeds, dedication and selfless sacrifice.
Padmini Arhant
Ukraine – Russia War Strategy – Padmini Arhant
Ukraine-Russia Permanent Peace Treaty 2022
Ukraine – Russia Peace Treaty 2022
Russia – Ukraine Peace Agreement
RUSSIA – NATO Permanent Peace Deal 2022
New Year 2022 Message – Padmini Arhant
India Republic Day 2022 Message in Hindi – Padmini Arhant
Padmini Arhant Skydive in California, USA, November 28th, 2021
Padmini Arhant and Roshni Greeted by the Brilliant Light in the Horizon

U. S. – South Carolina Senator Lindsey Graham
The U.S. Senator from South Carolina Lindsey Graham could exemplify sincere concern towards mankind by holding Senate hearing on the controversial gain of function research on corona virus, the fundamental cause behind the global pandemic.
The individuals and institutes in the United States such as Professor Ralph Steven Baric from UNC, Chapel Hill, North Carolina, Dr.Anthony Fauci, NIH, Investors Bill and Melinda Gates among others are accountable for the loss of millions of lives worldwide.
United States alone lost nearly million lives and thousands in serious health conditions till date.
Senator Lindsey Graham – please call on these members and subject them to public hearing under oath applying the same rule of law as ordinary citizens for their involvement in the worst human tragedy and health as well as economic disaster.
It is incumbent upon every United States lawmaker and duty to hold these members accountable for the crimes against humanity.
Senator Lindsey Graham’s call fior assassination of foreign head of state such as Russian President Vladimir Putin is irresponsible, interventional and unnecessary provocation.
Please focus on getting own house in order on the global pandemic responsibility.
Padmini Arhant
Superpower Quest
Those who are in the game to rule the world allowing their individual and collective minds to be ruled by negativity and counterproductive strategy is the biggest irony.
Those who have no control over dominant ill-vices control nothing including own will and reason.
Padmini Arhant
Evil – Definition and Reason
What is Evil?
Evil is sinful with malice, wickedness, violence, corruption and extreme criminality.
Why does Evil exist?
Evil exists predominantly due to fear transformed into silence among victims of evil actions.
From the days of crucifixion of Jesus Christ to assassination of leaders pursuing the path of peace, harmony and individual rights and the latest depopulation agenda via global pandemic, evil has unleashed wrath on humanity until now.
Although evil feigns power and proclaim itself the most feared among those evil dominates, the reality is evil is the biggest coward, cruel, weak, deceitful and dishonest element.
Evil is own enemy to itself. Evil invariably succumb to evil thoughts and deeds. Evil’s permanent abode is hell and accordingly engage as hell raisers on earth inviting apocalypse upon self and evil contingency.
Padmini Arhant
Global Pandemic Accountability
It’s high time all those engaged in the controversial Game of Function research and SARS COV2 caused global pandemic are held accountable for heinous crimes against humanity.
The researchers, investors, facilitators and promoters individually and as an institute as well as organizations profiteering from the catastrophic health and economic calamity are equally liable for irreversible deaths and devastation originating from the lab manufactured virus. Accordingly, they must be brought forward to accept responsibility.
Until then any decisions from them as moral authority against anyone on any matter is illegitimate and invalid.
One must be the example prior to expecting others to set an example. The international rules must be fair holding none above law on such matter barring exception especially when the crises is related to major health disaster in human history.
Padmini Arhant
Human Lifestyle
Human lifestyle
The human lifestyle beginning with food linked to beef controversy killings and mob lynching in India, traditional clothing like the current hijab rights in Karnataka, India or contemporary attire to suit personal preference to name a few are exclusive fundamental rights in a society.
The rules restricting one while allowing others to practice religious or cultural customs is discriminatory in a democracy.
The state and any other internal or external intervention defining and determining others personal life, identity and way of life i.e. customary beliefs in public or private is undemocratic, unethical and uncivilized in the modern age.
Humans regardless of race, religion and gender have the right to exercise individual discretion on their lifestyle in accordance with their personal choice barring any physical injury or public safety hazards.
Padmini Arhant
Satanic Age
In the satanic age, Satans insist one must engage with them in satanic terms.
What are Satanic terms?
Satanic terms are typically smothered in hatred, prejudice, misogyny, condescendence, divisiveness and hypocrisy.
Why do Satans always get it wrong?
Satans suffer from obsessive disorders. Once they are obsessed with anyone or anything, they pursue them or that endlessly for Satans are clueless. Their cluelessness and obtuse obsession invariably lead them to precipice and experience imminent mighty fall.
What is the reason behind Satans use of proxies, pawns and puppets to smear and attack their obsessed target?
Satans are essentially devils in disguise. Since the devils never deal with their fixated target directly due to cowardice and wickedness, they indirectly launch assaults against their enemy.
Above all, Satans are enslaved to evil actions and connivance. Accordingly they hire proxies, pawns and puppets, who in return are more than happy to be Satans’ slaves and dalliance with the devils.
Evil reaps what evil sows in life.
PadminiArhant
Clueless – Fact
The important clue on clueless is denial of reeking pseudo supremacy and uninhibited misogyny shattering false image and duplicity much to self-embarrassment and detriment in the present time.
Some things never change no matter what happens in the turn of events with glaring reflection on degradation of human value.
Padmini Arhant
Greatness Expression
Greatness is best expressed in selfless deeds and endless sacrifice more than any other characteristics defining human value.
Padmini Arhant
Definition of Fascism
What is Fascism?
Illegitimacy, Impotency, Intolerance, Incompetency, Inadequacy, Indecency, Ignoramus Idiosyncrasy.
Illegtinacy – in birth.
Impotency – Impotent in confronting exposure of Fascist rule.
Intolerance – To facts, TRUTH and ground reality.
Incompetency – Absymal performance.
Inadequacy – megalomania.
Indecency – Impropriety and inappropriate response to defend the indefensible position.
Ignoramus Idiosyncrasy – fascism indigenous.
Padmini Arhant
Pandemic Bulletin
WHO Director General Tedros Adhanom Ghebreyesus on the pandemic;
“If we end inequity, we end the pandemic,” said Dr. Tedros Adhanom Ghebreyesus.
In fact, the reality is otherwise.
The end to consistent misinformation, misguidance and mass confusion from the health authorities would lead to the pandemic end.
The directives and guidelines from WHO Director General since January 2020 followed by public health authorities viz. Dr.Anthony Fauci, NIH, CDC and other government agencies until now responsible for the status quo – the pandemic and variants prolonging the battle.
Padmini Arhant
India – Untouchability Atrocities Impetus Proselytism
India – Untouchability Atrocities Impetus Proselytism
The man made hierarchy in the social structure falsely attributing the abhorrent untouchability custom in Indian society as God’s will is primitive and pejorative.
The tradition initially conceptualized and imposed by brahmins – the foreign invaders’ descendants displacing the native population in Tamil Nadu, India – the origin of life and cultural bastion having established civilization long before the emergence of other continents on earth.
The brahmins declaring themselves the chosen ones to serve God adopted the title priesthood and placed their identity on the top tier in the hierarchy fraudulently citing the caste orientation as Vedic inscription. They conveniently placed the rest in the lower tiers of the hierarchy and exerted control over the religion they adopted upon arrival in India following the conclusion of Indus Valley Civilization. The word Hindu and accordingly Hinduism is a foreign named identity.
It has nothing to do with the ancient sacred spirituality presided by God Shiva – the Supreme Soul epitomizing the light, energy and sound encompassed as audible, visual and sensed energy within.
The sacred symbol and syllable OM originating at the dawn and creation of the universe epitomize the interconnectivity of light, sound and energy experienced in five sensory organs of sight, hearing, breathing, taste and touch.
The mantra OM is related in other religions as AUM and Amen in acknowledgment of the Soul within in connection with the Supreme Natural Phenomenon.
Similarly, the greeting salaam or As-salamu alaykum in Islam ending in OM sound and in Judaism the greeting Shalom has OM meaning Peace.
The hideous casteism in Indian society was sowed by foreigners as Aryans and Kazhar Ashkanazi Jews claimed as such by the brahmins in Indian society.
The duality of Aryan and Jewish traits maintained by them to sync in with contemporary International syndicate and win favors from all sides known as the Chanakya tactic. Chanakya – a controversial brahmin advisor in Chandra Gupta Maurya period in history.
The dravidian concept is yet another political spin from the notorious factions like Periyar and Dravida Kazhagam in Tamil Nadu posing as atheists, typically the alter ego fomenting and fostering brahminic ideology in exploiting religion and society for personal, political and vested interests.
The insidious prejudice practicing untouchability and outcasts until today were further promoted and institutionalized by the corrupt criminal and scandalous political class in India post-independence as they lack legitimacy in governance and usurp to power via rigged election as foreign puppets and proxy.
Padmini Arhant
Evil Shuns Light
Evil actions upon being brought to light is found annoying by those engaged in evil activities.
What a surprise?
Unveiling ugliness of those hiding behind glossy facade is necessitated by evil cursing the truth as deranged, when in fact deformed in character are those unabashedly abusing status depriving and denying others their legitimate rights.
The mirror reflection for evil is understandably unbearable.
Why not quit being evil and become human and humane for a change in the unknown and unpredictable lifetime?
No harm in trying and only gains upon renouncing evil.
Padmini Arhant
Warning – Intruders, Repeat Offenders and Violators of Individual Civil Rights
This message is strictly for intruders, repeat offenders and violators of personal rights, liberty and space.
In a real free world, exercising individual rights against serious violations is inalienable dignity.
If you are a peeping Tom from wherever currently or sever since my existence plagiarizing my hard work and identity attributing to anyone famous or unknown then read the following as well to be clear of the consequences of your deceitful actions, theft and despicable behavior.
Whoever is indulging at whosoever behest in intrusion of my home, personal life and exploiting me for more than a decade unpaid i.e. free labor, you have invited upon yourselves unnecessary irreversible excruciating pain, suffering and damages in many dimensions.
The unfortunate yet deserving karmic effects are your own doing with none to blame except yourselves and those whom you are obliging much to yours and their peril.
Any attempt to disregard this warning and continue to violate my personal rights, liberty, space and that of my family not shall but in fact will only deliver regrets and irreconcilable outcome.
Padmini Arhant
Free Speech Censorship
Free Speech Censorship
The videos shared on this website on Ivermectin and India’s PM Narendra Modi’s divestments from the Indian economy to offshore viz. Afghan infrastructure aping counterpart China’s Premier Xi Jinping’s Belt Road initiative were removed imposing censorship on free flow of information in public domain.
The videos have been re-published on a different platform.
Obviously the evidence based data are not acceptable to members and entities experiencing discomfort and indigestion on TRUTH and facts based information.
The supposedly powerful elements as political and health industry authorities have established their powerlessness via intolerance to Free Speech and substantiated information on science, economy and the corrupt political system disguised as democracy.
The real power rests in level playing field with equal opportunity and fairness demonstrating tolerance and respect for alternative views and voices that constitutes functional democracy.
Unlike the current trend silencing and enforcing cancel culture revealing the true colors of authoritarianism only expedite politics, science, economy, religion and politics controlled social media decadence imminent end.
Nonetheless, TRUTH will prevail and has since time immemorial against falsehood and fascism.
Padmini Arhant
Author & Presenter
PadminiArhant.com
Prakrithi.PadminiArhant.com
स्वतंत्रता v. बंधी
स्वतंत्रता v. बंधी
जो व्यक्ति बंधी हो – सत्ता, काला धन और सबसे ऊपर अहंकार, अभिमान और मूर्खता में दबे हुए हों फ़िर भी अपने आप को राजा समज बैठे तो उनका और उनको शेय देनेवाले से लेकर चापलूसी करनेवाले तक की कोई कल्याण हो ही नहीं सकता ।
आकाश में बादलों के बीच निडर उड़ने वाली पंछी अपनी स्वतंत्रता और स्वाभिमान को लहराती है । यह पंछी संसार के सबसे ऊंचे छोटीपर पहुँच कर पूरे सृष्टि रचयिता को अपनी शीश झुकाकर उनकी आशीर्वाद की योग्य बनती है तो वो कई सर ताज से अधिक मान्य है उसकेलिए |
भले ही दुष्कर्म और दुष्टता के बंधी और कार्यकर्ता उस पंछी को अपनी दुर्बल दृष्टि के कारण क्यों न देख पाते| यह उस पंछी के लिए कोई अंतर नहीं पड़ता |
लेकिन जो बंधी है हर तरह से – उन्हें जीते जी और उसके बाद भी बहुत पश्ताना होता है |क्यों की जिनका मन हमेशा दुष्कर्म में कैद हो और दुष्टता के चरम पर बैठे हुए हस्तियों के आदेश की सेवक बनजाए, तो राजा, महाराजा, शहंशाह या बादशाह का ताज़ पहने हुए भी वह मात्र सेवक और हारे हुए फ़क़ीर ही होते हैं |
पद्मिनी अरहंत
अंधेर नगरी चौपट राजा।
अंधेर नगरी चौपट राजा।
पद्मिनी अरहंत
जय राम जी की।
हुआ यूं की कुछ व्यक्ति अंधेर नगरी के सेवक बने। अंधेर नगरी काला धन और काली करतूतों के लिए ही प्रसिद्ध है। जब यह सच खुलकर सामने आया तो वे व्यक्ति जो अपने आप को प्रधान सेवक कहते फ़िरते हैं, हालांकि यह सेवक हैं पूँजी पतियों की और मुख्या का रूप धारण करते हैं केवल आम जनता पर रोभ जमाने के लिए। जैसे गरीब किसान, मज़दूर, आदिवासी, अन्य समुदाय के नागरिकों को पिछड़े वर्ग में दखेलकर और धर्म यहाँ तक भगवान का भी राजनीति और सौदा करते हैं। जब अंधेर नगरी की पोल खुली तो पूँजी पतियों के सेवक की कुर्सी हिलने लगी। यह तो होना ही था।
आख़िर सच और मूच ज़्यादा देर छिपाया नहीं जा सकता। यह कड़वी बात सच के विरोधियों को तनिक भी अच्छा नहीं लगता। ख़ैर उनके पसंद नापसंद को लेकर न हीं सृष्टि बनाया हैं न ही उनकेलिए यह संसार जुकता है।
फ़िर भी अंधेर नगरी के सदस्य और पूँजी पतियों के सेवक के ब्रह्म टूटने का नाम नहीं ले रहा है।
वैसे अपने छवी को लेकर आम जनता के ख़ून पसीने से कमाया राष्ट्रीय धन लूटाने वाले व्यक्ति, दूसरी और उस पैसे को आइना दिखाने वालों पर जासूसी के लिए खर्च किया जाता है |
यही व्यक्ति और इनके साथी की प्रतिक्रिया – किसी प्रकार के संकट के कारण दूसरे देश में शरण लेने वालों को गुसपेटि कहकर उन्हें वापस लौटाते हैं | उल्टा खुद दूसरों के घर बिन बुलाये पेगासस जासूसी के माध्यम गुसना ज़रा भी अपराध नहीं मानते हैं | इन गुसपेटियों के लिए कोई कायदा नहीं | औरों के लिए अलग खानून चलाने वाले किस प्रकार के लोग हैं, इसे बार बार अपने काले व्यवहार से दिखाते हैं |
और तो और इन्हें वृन्दावन में निधि वन के नियम का ज्ञात होते हुए भी, पेगासस के ज़रिये अपनी दुर्गति को आमंत्रण करते हैं |
जो व्यक्ति मुख्या के तौर पर आम जनता के सामने तरह तरह के पौशाक और पगड़ी पहनकर समबोदित करते हों, यहाँ तक करोना बीमारी के बीच चुनावी भाषण देकर कई लोगों को मृत्यु दण्ड देने वाले, जनता को अक्सर टोपी पहनाने की शौक़ीन भी हैं |
स्वयं पगड़ी पहने और जनता जनार्दन को टोपी पहनाने वाले व्यक्ति, अंधेर नगरी के चौपट राजा ही कर सकते हैं |
इस चरित्र को अपना सबसे बड़ा गुण मानते हैं और इनके टोली एवं पलटन इसे बड़े गर्व मानकर इनकी झूटी प्रशंसा करते हैं |ऐसे होते हुए यह व्यक्ति और इनके प्रशंसक एवं श्रोता न घर के न घाट के रह जाते हैं |
जय राम जी की।
पद्मिनी अरहंत
Social Media Censorship
Why COVID-19 ORIGIN Not a Major Concern for the Ruling Political Party?
Galactic Spectacle - Double Rainbow
Truthful Reflection
Changing the mirror makes no difference to image reflection.
Rejecting the truth sayer never make the truth disappear.
The one who knows it realize sooner than later the truth about truthful reflection.
Padmini Arhant
न घर के न घाट के – अस्तित्व दुविधा Identity Dilemma
न घर के न घाट के
–
अस्तित्व दुविधा
Identity Dilemma
पद्मिनी अर्हंत
जिन हस्तियां में अपनी गलती और अपराध मानने की न हिम्मत या नीयत हो, और वो बलि की बकरी पे अपनी घटिया ज़िन्दगी और कारनामायें तोपते हैं तरह तरह के अभिनय से – कभी नमस्ते, कभी जीत का इशारा, कभी काला चश्मा पहनकर – जैसे की पूरे विश्व में केवल एक मैं ही काला चश्मा या आँख का चश्मा पहनती हो।
और उससे भी बत्तर इनकी हरकतें बंदरों वाली नाच खेल करते हुए, अपनी हुलिया पर उंगुली उठाने वाली नौटंकी में व्यस्त हों, ऐसे बेईमान व्यक्ति अपने आप को मसीहा कहते फ़िरे, तो इनके मानसिक अवस्था को ही सामने लाता है।
यह लोग जिन्हें अपनी जीवन निपटने की हुनर और धैर्य नहीं, यह भला औरों की पाप का भोज क्या ख़ाक उठाएंगे? जो यह मसीहा बन पड़े!
अगर यह महोदय गण दूसरों को तंग और दुख न दे, तो यही इनके लिए और इनके जितने भी आस पास के सगे सम्बन्धी और पूरे समाज के लिए बहुत बड़ा उपकार होगा।
मगर बात ये है की यह लोग अपने आप से भाग रहे हैं। किसी और के अस्तित्व को अपना बनाकर अपने ढोंग धोखलापन को और निछावर करते हैं।
इसी में इनकी बेवकूफी और बेचैनी बेहाल सामने आता है।
बेचारे किस्मत के मारे। अपने आप को महान मशहूर यानी प्रसिद्द कहने वाले, कितने अँधेरे मैं है, यह बात इन्हें नहीं पता।
यह इनका दोष नहीं। इनकी अल्प बुद्धि का हाल है। ऐसे होते हुए, यह न घर के न घाट के हो जाते हैं। इन पर तरस के अलावा कुछ नहीं आता। जब इंसान को अगले क्षण का ख़बर नहीं, यह निकले अपनी ऐतिहास पलटने।
यह प्रयास इन्हें सिवाय निराश और अपमान के अलावा कुछ नहीं बाँट रहा है इनको, तब भी यह अपनी हार मानने को तैयार नहीं।
अब तक इनका कब्ज़ा धन, दौलत, शोहरत और सत्ता तक था। अभी यह लोग इज़्ज़त के ठेकेदार भी हो गए। इनको इतना तो पता होनी चाहिए की इज़्ज़त देने वालों को ही इज़्ज़त मिलती है। जो दूसरों की अस्तिव को अपना बनाकर, उनके व्यक्तिगत अधिकार छीनकर फ़िर उन्हीं को निशाना बनाने वाले, भला उन्हें ही इज़्ज़त की पाठ पढ़ाएंगे?
यही हुई ना बात – चोरी और ऊपर से सीना ज़ोरि!
कब यह अपनी गलती पर ध्यान देंगे? इनका तो यही मानना है की यह लोग कभी किसी को हानि पहुंचाते नहीं। किसी के साथ अन्याय कर ही नहीं सकते।
यह दूध और चन्दन से धुले हुए है। इनकी तरह कोई संसार में धन्य है ही नहीं।अगर यह नहीं होते तो यह संसार कब का समाप्त हो जाता। इनके पवित्रता से ही जीव जंतु प्राणी के जीवित रहना संभव रहा है। वर्णा इस कलयुग में मृत्यु, शोक और दरिद्रता के अलावा है ही क्या मनुष्य की जीवन में?
चलिए आपको हम सिखाते हैं की किसी के छोटेपन को ना बढ़ाने में अपनी बड्डपन है। आप भी क्या याद करोगे।
इनका भी जवाब नहीं। क्योंकि आज तक इन लोगों की ही मन मानी चलती आयी। तो इन्होने सोच लिया की इनका ही दौर सदियों चलेगा ।
जब रुत बदलते हैं, सरकार बदलते हैं, यहाँ तक की यह रातों रात काला धन से माला माल हो जातें है तो युग और समय बदलने में इनको क्या संकट है?
आख़िर इनका इस दर्ति पर उतना ही समय है जितना अन्य जीव प्राणी का है। कई तो अपने जीवन के सफ़र पूरा नहीं कर पाते। ऐसे होते हुए इनका रोब और हुखुम जिन पर जमाते हैं जैसे की दूसरों को कुचलना इनका जनम जन्मांतर का अधिकार हो।
पृथ्वी हर 365 दिन में एक बार सूर्य की परिक्रमा करती है और हर 24 घंटे में एक बार अपनी धुरी पर घूमती है।इसके कारण २४ घंटे ४ समय – सुबह, दोपहर, शाम और रात में बटा हुआ है।
यह प्रकृति के नियम है जिसे किसी के लिए भी बदला नहीं जाता। जिन्हें भी इस प्राकृतिक के परमपरा से समस्या हो, उन्हें इस दर्ति में अनेकों बार जनम लेकर इस वातावरण का आदी होना पड़ता है। ज़रूरी नहीं इन्हें मनुष्य रूप ही प्राप्त हो।
वाह रे मसीहावों, आप लोगों ने हद पार कर ली आपके इस प्रदर्शन से।
आपको स्वयं पता नहीं आप है कौन?
और ऊपर से कोरोना का भी असर हुआ है आप मसीहा पर। शायद कोरोना को नहीं पता की आप लोग मसीहा बनकर घूम रहे है। भला कोरोना को आपके मसीहापन से क्या लेना देना? इस मामले में कोरोना बिलकुल निष्पक्ष साबित हुआ है। फ़िर भी यह जीवन है और यही आपके रंग रूप है।
इसे विलम्बना कहें या विनाश काले विपरीत बुद्धि ?
आपकी शुभ चिंतक
पद्मिनी अर्हंत
New World Order (NWO) Orchestra
What is the link between the pandemic and Great Reset?
Global economic shut down.
Pandemic forced economic lockdown and Great Reset in world financial system designed global economic shut down synchronized in the New World Order (NWO) orchestra.
Padmini Arhant
Free Speech from the Silicon Valley
Free Speech from the Silicon Valley
Padmini Arhant
The reference Free Speech from the Silicon Valley from paid anchor at The Hill serving as mouthpiece for those with acute indigestion to factual information and truth is cynicism ad nauseam.
As a matter of fact, Free speech not only from the Silicon Valley, Free Speech from anywhere is inalienable right of any individual in Almighty God created Free World.
However, the concept of free world is disseminated out of context limiting to those obsessed with reining control over others’ health, life and rights expecting herd mentality at large.
Furthermore, questioning anyone in power and those exerting authority via influence and dominance severely affecting life at individual or collective level is also an embodiment and essence of Free Speech. Again, one need not be elected to power or approved by the ruling class to exercise the fundamental right as obtusely asserted by The Hill propagandist.
This freedom presumptuously claimed as exclusive privilege and patented right of the so-called elected to political position or influential in society, who by the way abstain from their political responsibility and legislative duty under constitutional oath let alone moral obligation due to collusion and complicity with violators of individual rights and life on all matter.
Such tradition impetus free speech in view of concerns related to policy and actions with possible impact on family and community. The mere exercise of Free Speech seeking clarifications from scientific, world health authorities or political class evading transparency and accountability as entitlement is not a crime or forbidden act in a system propagated as democracy.
Importantly, not all elected are genuinely elected to power by people when election is increasingly a formality with actual votes whether 74 million or 700 million discarded to satisfy unruly system in the rigged electoral process.
Padmini Arhant
Original v. Fake
The differences between original and fake are;
The original is meaningfully true, natural and authentic whereas the fake is false, manufactured and disingenuous.
Those using the fake to justify their fraudulence convince the gullible on anything not the alert and wise.
Original is and will always be original. Imitation is an enamel that slips and fades after minimal use.
The example is real gold i.e. original and the imitation that is plated with artificial alloy painted in gold color. Needless to say there is no match in quality and value.
Similarly, the imitation could be embarrassing for those wearing wig blown away in public with gusty wind exposing reality.
Furthermore, the original need not prove anything to anyone and apologize to none for being as such – truthful and genuine.
Again, using the fake to bash or tarnish original highlight lame wicked cowardly tactics affecting the source and catalysts not in the least the original target.
Padmini Arhant
Life is a Gift
Life is a gift. Living own life is an art. Those who live others life i.e. wasting their time in snooping, spying and snitching on others typically declare themselves corpses for they never live own life or mind their business.
Get over envy before this dreadful disease called jealousy consumes you. Envy is also a sign of inadequacy and insecurity.
Malice is a malignant cancer that affects the source rather than the target exemplifying what goes around comes around.
Hatred and prejudice towards anyone is a reflection of inner self – the unhappiness and hopelessness in oneself evolve into unnecessary agony for them. Once again a self-inflicted injury eventually hurt the origin not the one that is aimed at.
Those who fail to live their life and instead steal others life and identity are morbid losers. They are ashamed of admitting their personal profile and characteristics while being foremost in targeting those who care less about them or their opinion.
With unknown factors on life existence such as time, condition and situation, don’t throw away life in obsession about anyone or anything that you can never impact except yourself. Such living is counterproductive and clearly signify lifelessness.
Life is a gift Live your life and let others live theirs.
Padmini Arhant
Enemy Profile
The enemy unable to fight own battle within self and succumb to self-destruction is least threatening and unworthy of any attention.
Could an enemy taking marching orders from Saitans within i.e. own mind and outside be smart, courageous and powerful?
Obviously not. Instead they are dumb, weak and powerless.
Could an enemy using proxies, pawns and puppets in nuanced attacks and insinuations have any merit and credibility?
The fact they rely on manufactured concoctions much to self-embarrassment and self-defeating purpose suffice response. Not to mention the inability to face whom they target via ethics and intellect, the attributes oxymoron to them except developing mob mentality.
The enemy utilizing own dead or alive inventory against the target exemplify fait accompli.
More than anything, the morbid culture to associate target with deceased exalt the challenged to glory in repeat resurrection to life compared to Jesus Christ in one time resurrection.
Could an enemy’s obsession and chronic fixation gain anything other than wasting life?
Such indulgence invariably result in self-mortification.
Why worry about an enemy preoccupied in self-termination?
Padmini Arhant
Filthy Swamp
Filthy Swamp
The Indian criminal political class hired peddlers nuanced podcast to attack facts and truth amplify the braying donkeys and oinking pigs in the swamp representing the filthy swamp.
To those podcasters in the caste oriented and religion baited nonsense, don’t indulge in anything or about anyone without complete factual information and knowledge about those for which you are paid to target via insinuation.
The Indian and supposedly Indian Americans (?) podcasters bragging about their nativity as Californians are reminded not to presumptuously assume exclusivity in this regard.
Personally there is not only a present native Californian in my family, the origin in California goes back long before California became part of the United States territory and Union – September 9, 1850.
Furthermore, my father as a Senior Executive in foreign international airline – British Airways visited the states California, New York and other parts of the United States back in 1950’s onwards and later.
In my view 1950 precedes 1960. Not to mention my original ties with California.
Never take anyone for granted to appease the corrupt and unscrupulous.
‘You are whom you represent in life.’
Padmini Arhant
Fait Accompli
Dealing with fait accompli is always hard for the side hoping and betting on fraud and crooked means to gain power.
The election 2020 is fait accompli for candidacies exposed on massive election corruption.
Padmini Arhant
Facebook, Twitter Accounts
The information regarding my account with Facebook is false.
“Padmini Arhant is on Facebook. To connect with Padmini, join Facebook today.”
This is FALSE. I am not on Facebook and never used the account since the social media inception.
Twitter – I quit twitter at the very onset of Twitter in public domain almost a decade ago.
Twitter prohibited my tweet for defending ordinary retailers, small business owners and particularly street vendors in India, whose livelihoods were targeted by then President Barack Obama and wife Michelle Obama during their trip to India to promote the world’s largest retailer Walmart in Indian market.
The retail giant Walmart entry in Indian market was a direct threat to millions of small retailers and shopkeepers whose only means of survival was the local marketplace in every nook and corner of India that was aimed at by globalists using political influence.
Twitter action was to comply with free speech restriction from Barack Obama administration targeting anyone voicing concerns over Barack Obama’s policy and plans detrimental to population in the United States and abroad.
Michelle Obama, the Board member of Walmart was actively engaged in canvassing for the retail giant during Barack Obama’s official term in office despite conflict of interests, a major objection for the media and democrat party against successor Donald J. Trump when sworn into office in 2016.
Unlike for the Obamas, the media and democrat party demanded incumbent Donald J. Trump relinquish personal interests and rights in Trump Estate and Trump business empire apparently obliged by latter at that point in time.
Padmini Arhant
Free Speech
Silencing Free Speech is Tyranny.
Forbidding Free Speech is FEARING TRUTH.
Freedom is non-negotiable inalienable right.
Padmini Arhant
Corrupt News Duplicity and Treason
Corrupt News Complicity and Duplicity
Padmini Arhant
Attention: CORRUPT NEWS FYI
Your contradictory position pretending to present China spy gate and purported cyber attacks from China against United States while promoting China funded lackeys to power is duplicity at best and treason at worst in credibility.
The real enemies like corrupt news in collusion with communist regimes and foreign infiltrators, the nation need to look no further than within to drain the swamp.
Corrupt news – your complicity with authoritarianism might be exclusively for pecuniary interests.
However, the betrayal of citizens trust aimed at subjugation and surrender to foreign authoritarianism such as China CCP and others at the cost of individual freedom and sovereignty is persona non grata (UNWELCOME).
Those who cherish independence in a free country and wish the same for others to be a free world would never acknowledge nor appreciate any threats to inalienable rights and liberty.
Padmini Arhant
Padmini Arhant Identity Clarification
Words of Wisdom – Padmini Arhant
Argument with fools is denying self-intelligence.
Truth is spoken once with clear conscience.
Lies spun in the web of lies strangle the liar.
The guilt promotes perpetual lies constricting courage and integrity.
Padmini Arhant
Mind Your Affairs
Mind Your Affairs
Padmini Arhant
Minding own affairs is the place to start to get things in order.
Unfortunately, there are folks preoccupied in invasion of others’ space and rights much to own peril. They waste life that could be lived productively in otherwise counterproductive lifestyle.
Checking own shortcomings and status quo for improvement and revival from point of no return would provide basic guidance in averting disaster.
Fatal obsession with others life and identity is destructive. It leads to indefinite suffering and burden on soul. The agonizing experience guaranteed upon departure from the world and beyond.
Live your life not others in the limited life span. Life is an opportunity to deal with karma. It is continued until account settled with dues to none.
Any satisfaction from hurting and harming others is sadistic pleasure. The indulgence internally impact source sustaining injury and insult to intelligence.
Never assume relief or redemption from sins and wrongdoings without penance and repentance.
Axis of evil with malice and hubris ends in the ditch.
The digger digging grave for others leaves the digger buried in return.
Nothing is free in time and space yielding profits and losses based on investments and strategy requiring ingenuity and labor. To some crooked means riding on others back and exploitation is inherent trait.
In following the simple rules, one could benefit living a fruitful and respectful life.
Thank you.
Padmini Arhant
Deep State Globalists “Black Lives Matter” BLM / BBS) Debunked
Deep State Globalists “Black Lives Matter” (BLM i.e. BBS) Debunked
Since when Fox News media mogul Rupert Murdoch from Australia become concerned and interested in Black lives?
Fox News anchors and hosts are good examples of how many ‘blacks and people of color’ are represented in public appearance and public relation jobs on globalists deep state television network.
The organization Black Lives Matter exploiting blacks and ethnic demography aimed at polarizing diverse vibrant society in the United States and other western nations like Britain, Canada, Australia where BLM/BBS synchronized reaction upon BLM violence on American streets unfolding in recent memory hardly escapes attention.
The stereotyped organization “Black Lives Matter” suggestive in name only is paradoxical in response to all lives including black lives generational suffering inflicted by globalists agenda to undermine ordinary lives across the spectrum for indefinite dominance as the privileged ruling class in the world. The status quo challenged visibly rattles the secret underworld.
Last but not the least the Fox News Corporation Murdoch heirs bribery to Biden campaign in the officially known investment of $1.1 million while the undisclosed funding kept unknown to sway democratic Presidential election 2020 in the United States prominently display the Fox unable to hide in sheep’s outfit anytime.
Again the indulgence in exclusive pecuniary interests at the legal votes and electorate expense verify the FOX natural instinct.
It’s time to reject globalists deep state propaganda and lackeys’ divisive identity politics.
Padmini Arhant
United States – Inconvenient Truth
United States – Inconvenient Truth
Welcome to Political Interpretation.
When a democrat hailed a winner, apparently it is
PROPHETIC
Juxtaposed, incumbent winner in 2020 is a MYTH?
Perhaps the New York Times could shed light on this political interpretation on equal but biased treatment.
Democrats surrogates – media and press and foreign financiers interpretation of election win and loss is;
‘Unless we are the winners by any means, anyone else deemed illegitimate.’
Those daring status quo are challenged for the entire term as demonstrated in 2016 until 2020 and now the same replayed in 2020 is hardly a surprise.
Precisely POLITICO, HUFFPOST, VANITY FAIR, PEOPLE, VOX, BUZZ FEED, THE WASHINGTON POST, NEW YORK TIMES, MSNBC, CBS, ABC, NBC, PBS, NPR, CNN, FOX NEWS… offshore media clique serving China CCP and GEORGE SOROS interests to delegitimize 74 million American votes is a travesty of democracy.
STOP CONTROLLING PEOPLE’s LIVES AND CHOICES IN A DEMOCRACY.
Human character defined in accepting victory with humility and defeat gracefully at any time.
Padmini Arhant
Delusion
A decorated ass will never be a horse except becoming member of braying donkeys and oinking pigs in the swamp.
Anything to the contrary is nothing more than delusion.
Padmini Arhant
Democracy Definition
What is democracy?
Ir’s not just about the government of the people, for the people and by the people. Instead it is installing the government of the deep state, for the deep state and by the deep state.
The deep state lackeys hired for a price and rolling over for positions deceiving those believing the massive deception as democracy is the irony.
In real functional democracy, tolerance and respect for others rights and their inalienable liberty to exercise discernment would be the norm eliminating identity politics and carte blanche authority to demonize whoever disagrees with them.
The resurrection of democracy is directly linked to deep state departure.
Is it likely to happen?
With greed and prejudice at the helm, the likelihood is expedited by those eager to succumb to own negative vices.
Padmini Arhant
Conspiracy Theory Era
Whenever incontrovertible TRUTH is evaded as “CONSPIRACY THEORY”, the forces and catalysts together with facilitators in media and networks airing the message bear the burden of lies and deception and ultimately succumb to own folly and fraudulence.
It’s not just the question of United States, it is a matter of lives on the entire planet forced to pay the price for willful egregious decisions with none accepting responsibility whatsoever.
Above all, the Federal Election Commissioner is expected to stay neutral and impersonal not become the hired representative of those subverting democratic process. All the more attracting accountability in any functional democracy.
Padmini Arhant
Words of Wisdom from Venerable Gautam Buddha
The Comedy of Errors
When the crew and contingency deployed to consistently lie and deceive public, they fail to see their desperate indulgence laughing at them. The sins and crimes committed to gain power drags them to bottomless pit much to their bewilderment.
The Zion centric, communist socialists, the pseudo progressives and the supposedly capitalist in collusion typically the deep state dark forces cozy secret society is rattled knowing the TRUTH that nothing lasts forever including their egotistical pathological obsession for dominance denying the overwhelming majority across the globe the inalienable right to freedom, individual rights and equal opportunity to peace, progress and prosperity.
The comedy of errors in all of this is;
61 + 47 = 100 (since when?). Even a kindergartener knows the right answer.
Apparently, these forces are extraordinary so they change the empirical mathematical results to their choice of outcome as they demonstrate in election.
According to those referring to self as a ‘Man of Faith’ notably faithful to unscrupulous and nefarious order citing a survey that 61% Americans think that incumbent President Donald Trump should concede to Beijing Presidency and,
At the same time quoting the same survey that 47% disagree of which 30% are democrats leaving only 17% republicans to disagree being eliminated in the Presidential race?
The election outcome on November 3rd, 2020 boasted approximately 150,000,000 (150 million) having voted of which 74 million is allocated to incumbent President Donald Trump which is 49% barring voter fraud in the contested election.
The propaganda alluding to 49% republicans who voted for Donald Trump are in agreement to delegitimize their legal votes while they are being further diminished in the fraudulent election and fabricated survey to mere 17% in the far fetched analysis.
How about the survey pointing out 30% democrats of that 47% in disagreement to incumbent President Donald Trump conceding to Beijing Presidency?
Does that mean in the projected win 51% to Joe Biden candidacy only 21% believe in dominion and media produced election result since 30% democrats do not concur with the outcome?
One need not be a math genius. The comedy of errors only reflect the conspicuous rigged election pushed forward as the historic win for Beijing financed and promoted Biden Presidency.
Yours Truly,
Padmini Arhant
Reaching the Mountain Top – Himalayas Mt.Kailash
Democracy on Decline – Padmini Arhant
Democracy on Decline
When Government run press and media focus on deflecting public attention from real issues.
Prime Minister assume priest position at the temple ceremony.
Prime Minister withhold reality from people on national security regardless of enemy dangerous aggression in the higher altitude Himalaya.
Zero tolerance to peaceful dissent and individual freedom.
Padmini Arhant
Life is a Blessing – Padmini Arhant
Life is a Blessing
Quit resolution on never to review, realize and reform ways with no remorse or regrets for persistent wrongdoings and harmful indulgence to hurt others.
Alleviate burden of sins while living rather than exacerbate in intentional violations.
You are who you choose to be despite life being an opportunity to live and leave in grace rather than disgrace.
KARMA is DHARMA ( Justice).
Padmini Arhant
Intellectual and Ethical Bankruptcy
Those in public service exploiting taxpayers with undeserving benefits and privileges are obligatory to accountability on their colossal failures rather than evading scrutiny.
Personal attacks, invasion of others privacy i.e. home and life ominously confirm the compulsive offenders intellectual and ethical bankruptcy besides their imminent end freeing the world from tyranny and injustice.
Padmini Arhant
Secret Society Demise – Padmini Arhant
The idea of secrecy and secret society installing puppet regimes as heads of states to implement anti-humanity agenda is directly responsible for generational human suffering and environment peril.
Secret Society end is the only way for humanity unity, peace, progress and prosperity.
Rejecting Secret Society and Deep state operatives from top to bottom in politics, media, economy, religion and society at large is fundamental for humanity liberty held hostage in the discreet clandestine meetings colluding against humanity.
No more Secret Society and Deep State authority under the guise of democracy and other form of political system.
Secret Society demise is elimination of evil and birth of the real free world.
Padmini Arhant
How to Change Status Quo? – Padmini Arhant
How to change status quo?
1. Stop listening to and following devils and renounce being the devils’ advocate.
2. Respect others rights as you expect to be done to you. Never claim anything in material or identity that is not yours and will ever be regardless of subversion and manipulation.
3. Shed your ego, arrogance and prejudice of all kinds in an effort towards self- cleansing and soul-searching leading to clarity on the importance of life not just yours but also others near and far.
4. Be yourself. Don’t engage in falsehood and fraudulent act.
5. Have courage and integrity to admit failures and accept responsibility.
Padmini Arhant
GREED – Padmini Arhant
Greed enslaves human mind directed at self-destructive course. Those consumed by greed experience misery in living and upon death. The actions prompted by greed invariably lead to tragic end. Greed is worse than any deadly disease inevitably terminating the source and all those influenced by the negative trait. Similarly infringement on others rights, illegal claims and usurping in any manner is carcinogenic and definitively cause unmitigated disaster for all those involved in the preventable malfeasance.
Result – End up losing everything including anything held before.
Remedy – Refraining from anything that never belonged to them and will never be theirs despite asserting sly woeful dominance.
Padmini Arhant
Life – Padmini Arhant
Life is a blessing. Live your life not others and introspect within to make it better.
Success granted denying others their legitimate rights to success is never a success.
Never claim anything that does not belong to you.
Repeating a mistake is blunder
Even the rock is reduced to rubble. Might and power are perceptions in the mind susceptible to folly and fallacy.
Be true to yourself so you can be true to others near and far.
When you seize others rights, You cease to exist.
Don’t run away from you. Face your inner self with courage and dignity.
Share love, peace and kindness towards all. Spare others from hate, envy and abuse.
Treat others the way you want to be treated by them.
Respect others’ life, rights and space.
Respect is a two way street. Not one way with a dead end.
Defeat your enemy within, you win love all around.
When others define you, that is impression. When you define yourself through your inner abilities and deeds that is self-realization – Nirvana.
Author
Padmini Arhant
Choice is Freedom – Padmini Arhant
Choice is Freedom
Free world and free society would represent freedom of choice.
Being able to exercise freedom in life on all matter define liberty.
None have the right or authority to seize others freedom in the space and time exclusive to every individual.
Forcing anything by coercion, manipulation or mass deception only result in resistance and rejection.
Choice is fundamental to distinguish one from another.
Merit translated into delivery quintessential in evaluating performance.
Substance important and relevant than symbolic synthetic symbiosis sponging off others.
Submission to compulsion is contradiction to selection and option.
Choice is the ultimate choice in living a life without having to pay a price in self-sacrifice.
Author
Padmini Arhant
Reality – Padmini Arhant
Lip service is common.
Delivery exemplify action.
Fake and imitation are not original.
Padmini Arhant
Saving Grace – Padmini Arhant
When your mind, heart and body always engaged in good Karma,
Your Soul experiences a home run!
Padmini Arhant
Free the World from Secret Society – Padmini Arhant
The world was created for all species in natural domain to be free and live life on their terms i.e. exercising individual freedom, experiencing equal rights and empowerment with fair opportunity.
The parallel world enforce entitlements claiming privilege, superiority and ruthless dominance despite self-worthlessness and sordid characteristics defining personal status.
The world deserves free, fair and open society.
Not false, fraudulent SECRET SOCIETY.
Padmini Arhant
Attention: Secret Society
Attention: Secret Society and Representatives
No matter how many fakes and impostors you produce to stymie my authenticity and identity as a woman of ethnic background, mother, great leader, honest and honorable humanitarian besides other noble attributes which I mention with humility that has positively served many from different walks of life.
Unfortunately, with the exception of those having exploited the same for their exclusive benefits driven by their negative traits and trend not without consequences in reaping what one sows in life.
Anything and everything you do via falsehood, propaganda and manufacturing phonies, proxies, puppets and pawns tied to unscrupulous, nefarious and criminal activities continue to backfire at you and them with many having met their Karma for their involvement.
The Sun rise in the East and your persistent efforts to sabotage natural cosmic event proved counterproductive many times over. Not to mention your folly and ignorance leading you to self-defeating cause.
Get a life. Live your life to make it meaningful and purposeful for you and you alone are responsible for your deeds and sins that you must settle sooner than later for any redemption and salvation which at this point in time is dismal and even impossible.
Thousand lies and million clones never replace indomitable TRUTH merged with the ABSOLUTE.
Truly,
Padmini Arhant
Divine Goddess Durga
Global Pandemic and China (Original) – Padmini Arhant
Corona Virus Pandemic Management
Corona Virus Management – Padmini Arhant
Corona Virus Global Pandemic – Padmini Arhant
Tamil OM Symbol & Pranava Manthiram
Absolute Truth

Reunion with Divinity God Shiva at Holy Mt.Kailash 2019

Padmini Arhant interaction with God Shiva during Parikrama of Mt.Kailash
Middle East Peace Plan 2020 – Padmini Arhant
Padmini Arhant Kailash Lake Manasarovar Pilgrimage 2019

Padmini Arhant on Parikrama or Circumambulation of Mt. Kailash
ARHANT Family at Famous Taj Mahal

Gautam Buddha’s Words of Wisdom

Global Economy & Nuclear Status – Padmini Arhant
Black Money, Corruption Culture in Politics – Padmini Arhant
Photo Gallery
Peace Pledge


Asunto Latino (Española)- Latino Affair (SPANISH)
Latino Affair – English
Vishwa Kalyan Award (VKA) 2018 Ceremony
Paradoxical Faith & Sai Baba – Padmini Arhant
Eliminating Casteism and Untouchability in India
Monkey See Monkey Do
Rude Awakening – Author & Presenter Padmini Arhant
India – Black Money in Election 2019
Nonsensical Trend
UP BEAT. By Padmini Arhant
World Classical Music Ensemble. By Padmini Arhant
 PadminiArhant.com
PadminiArhant.com